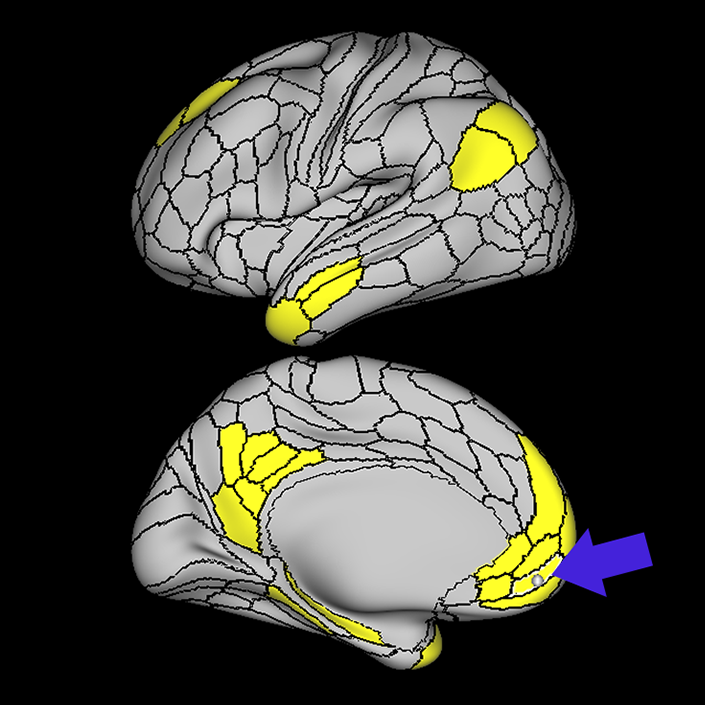
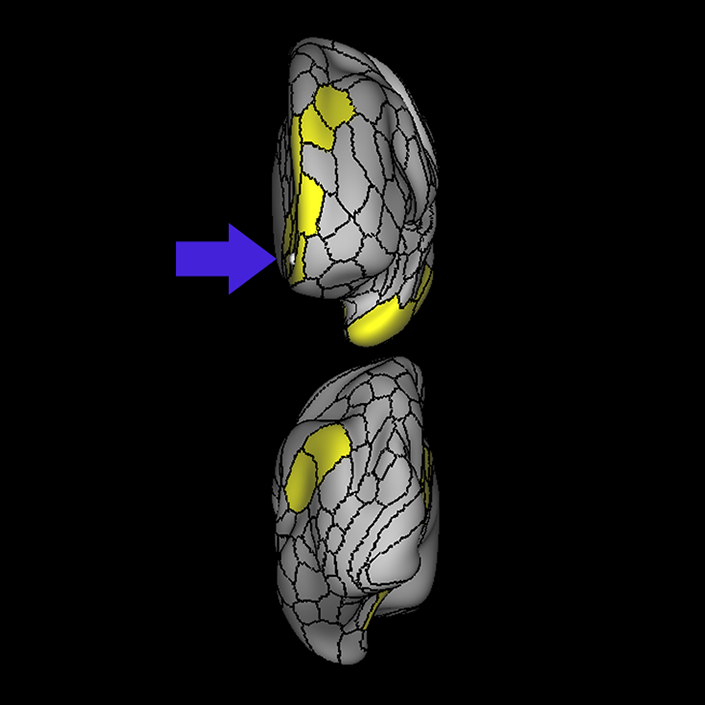
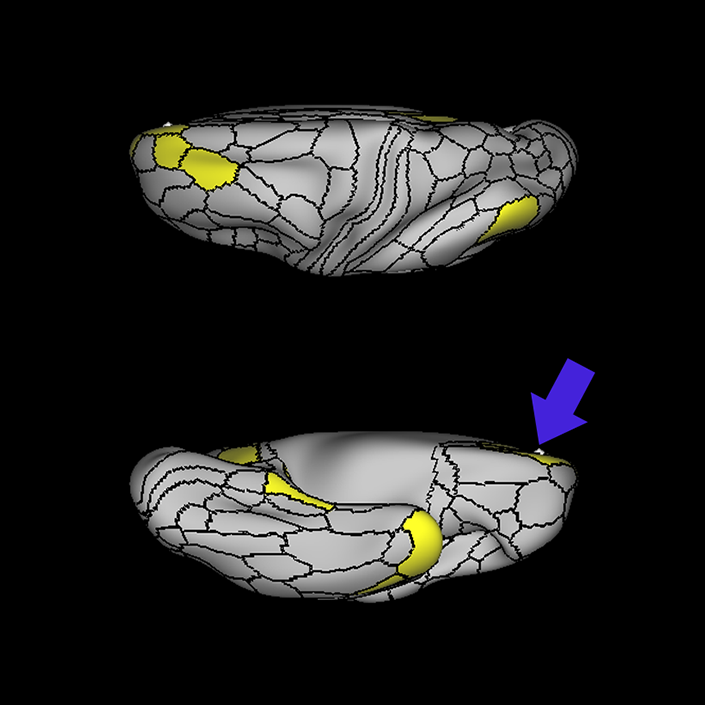
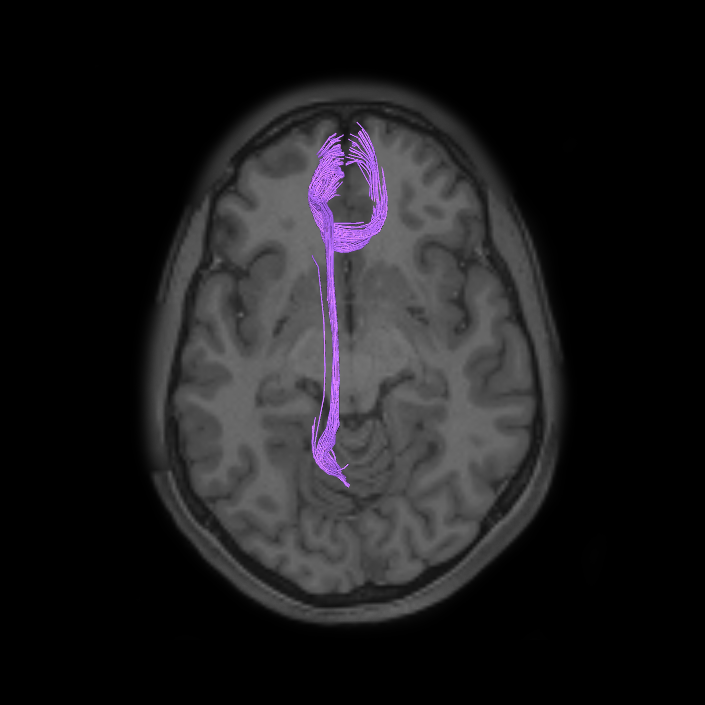
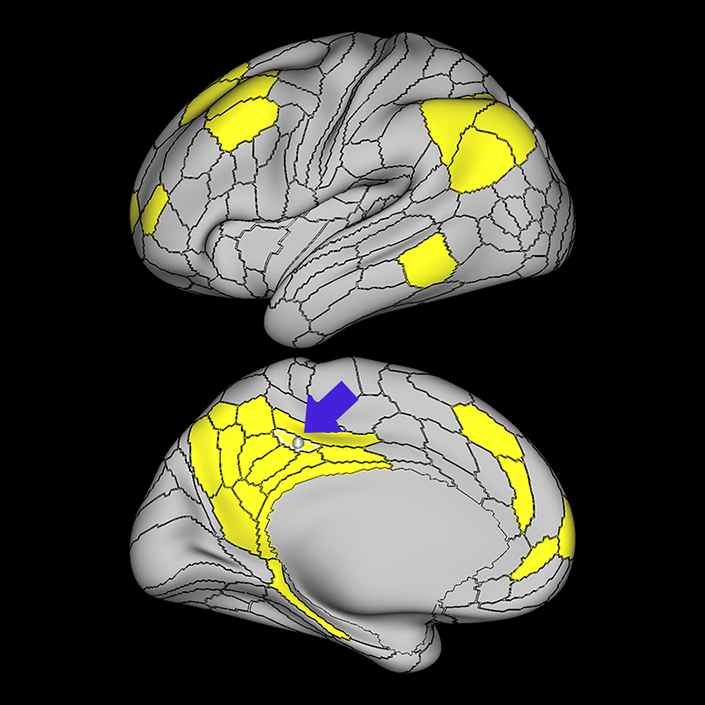
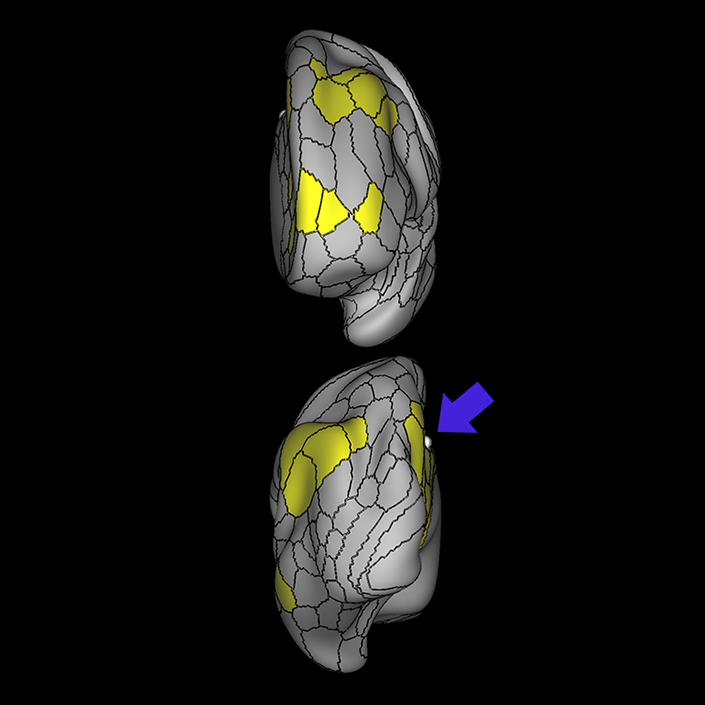
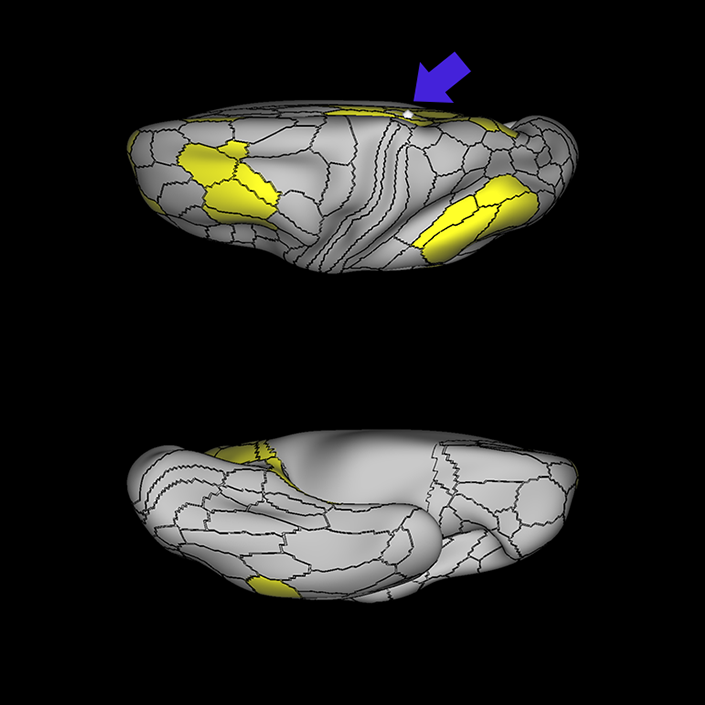
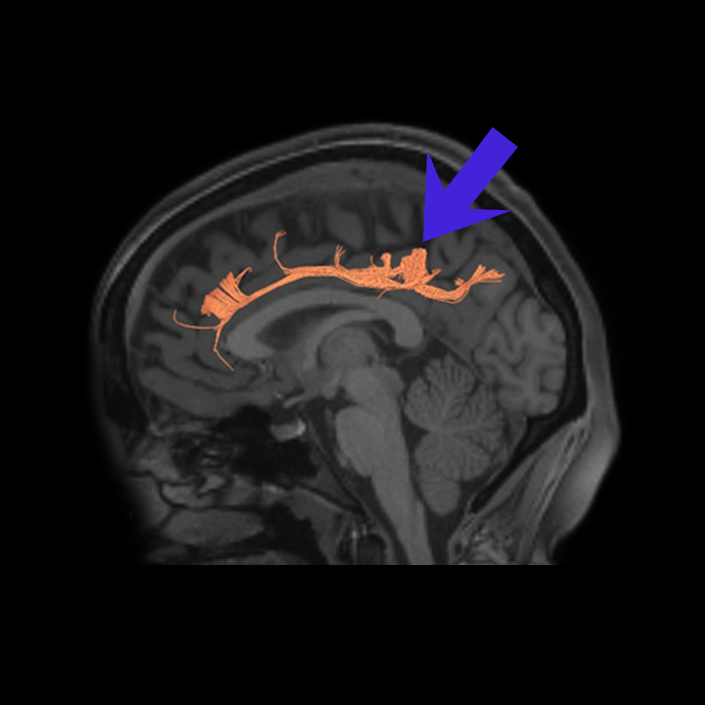
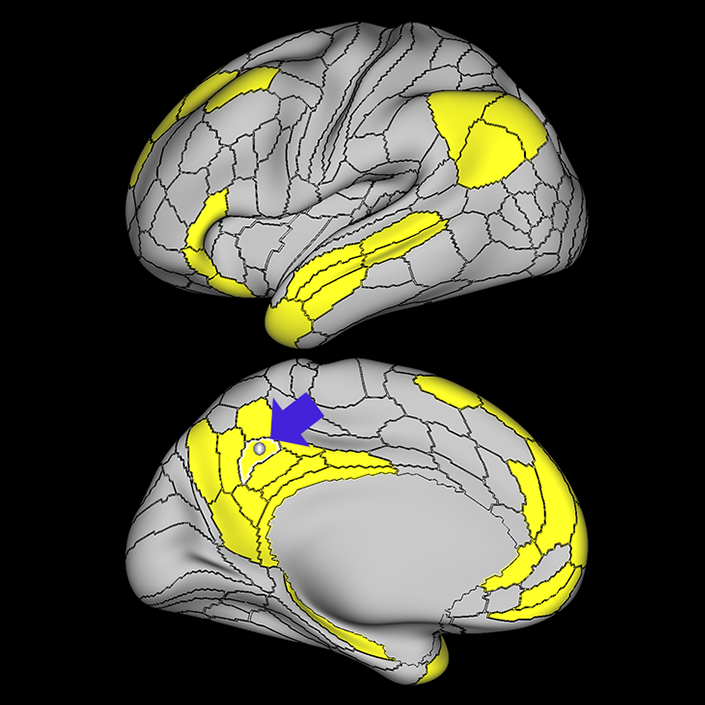
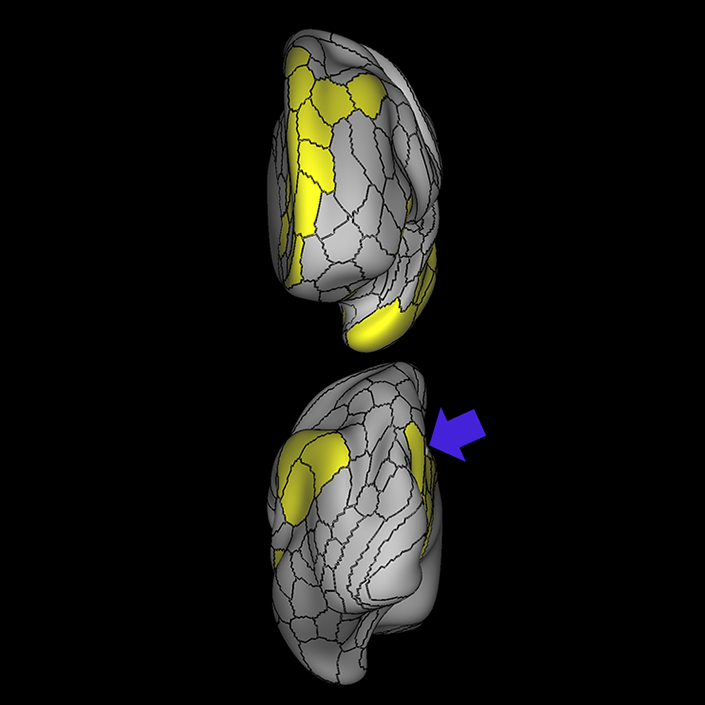
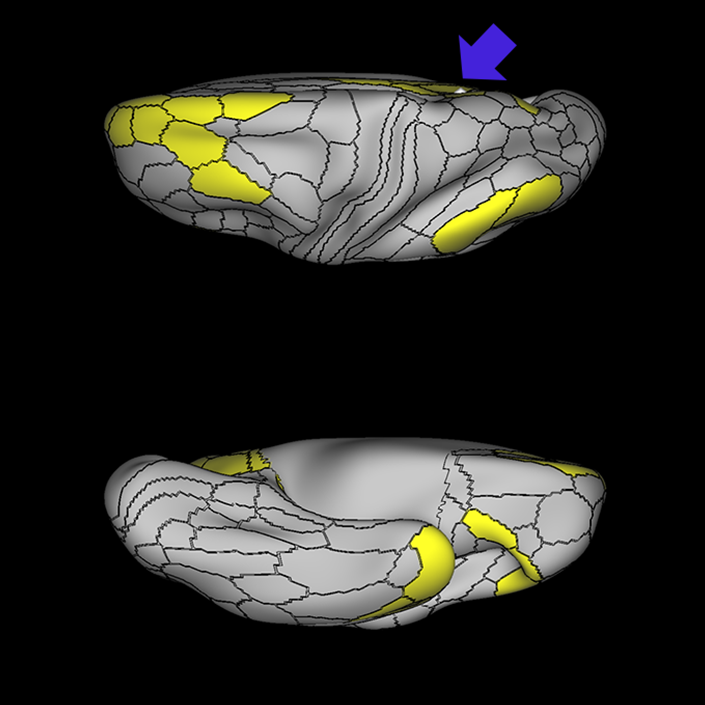
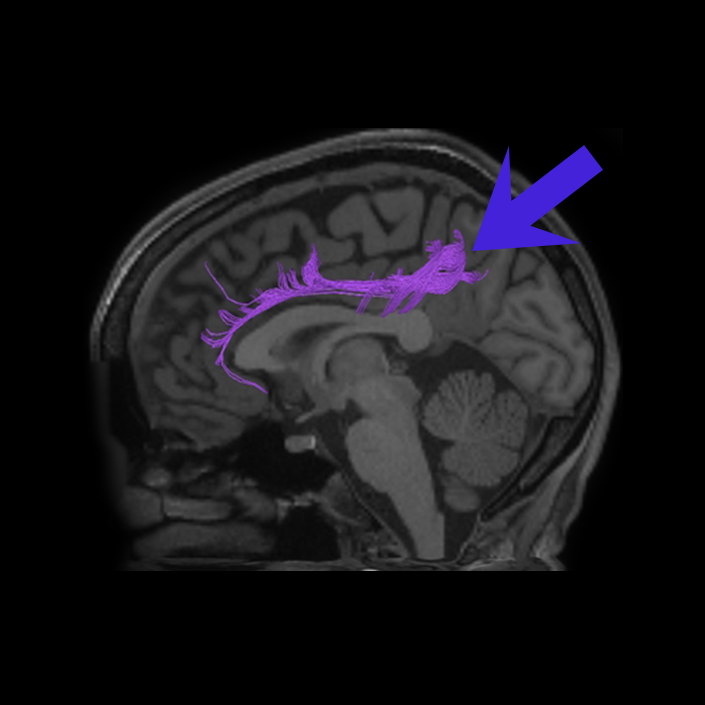
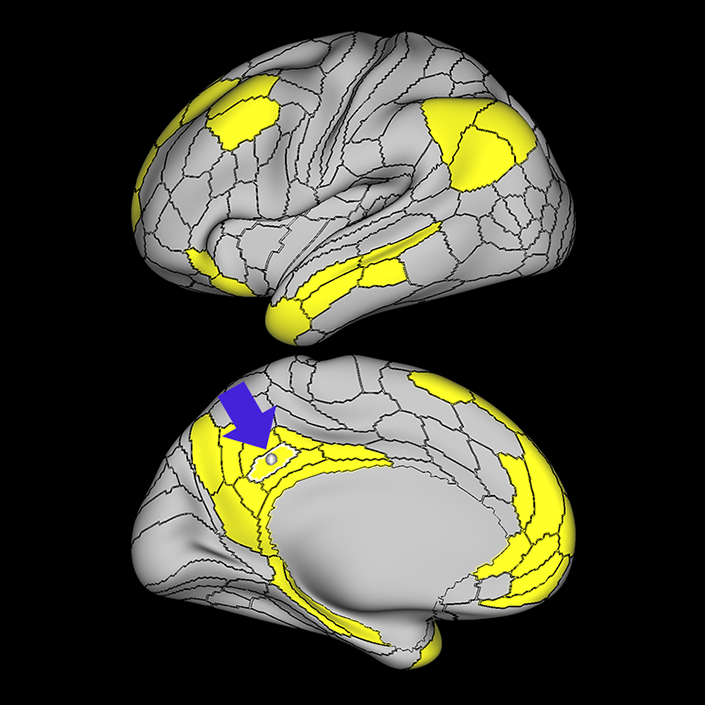
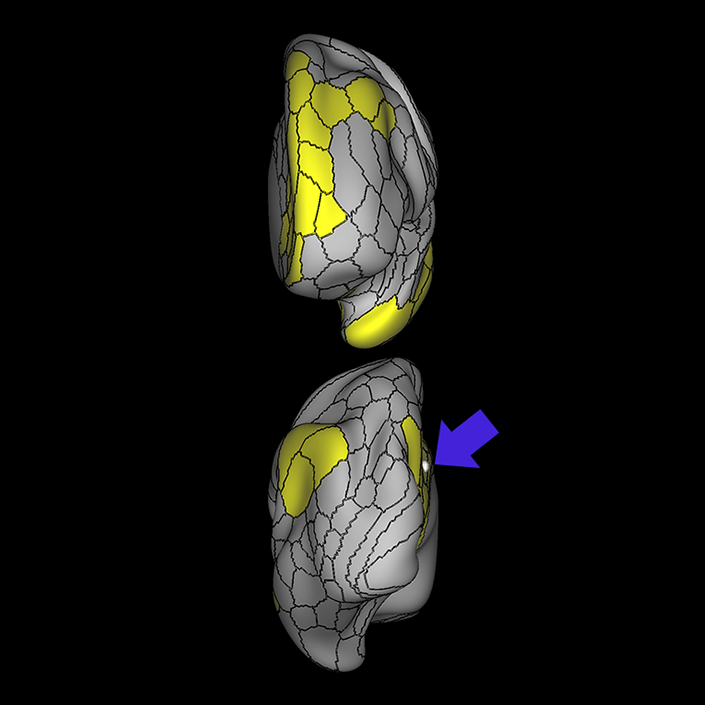
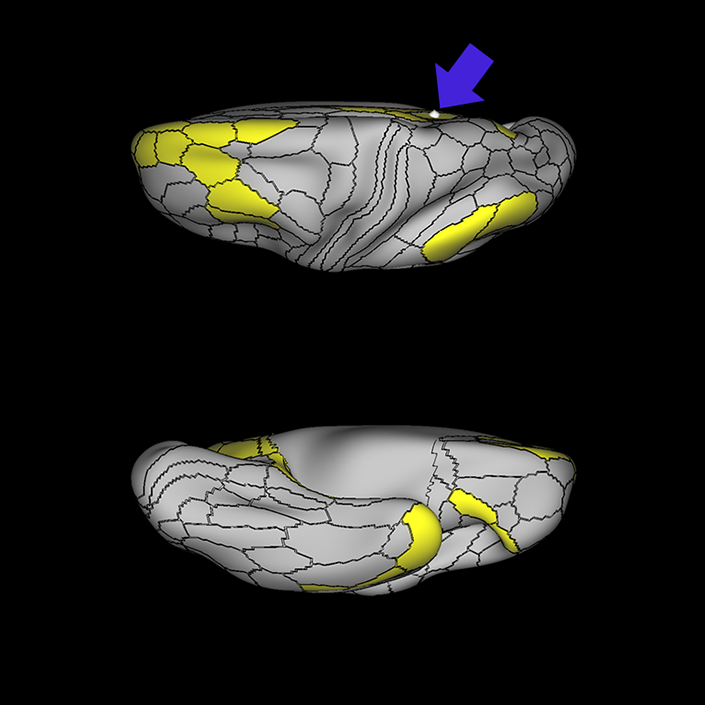
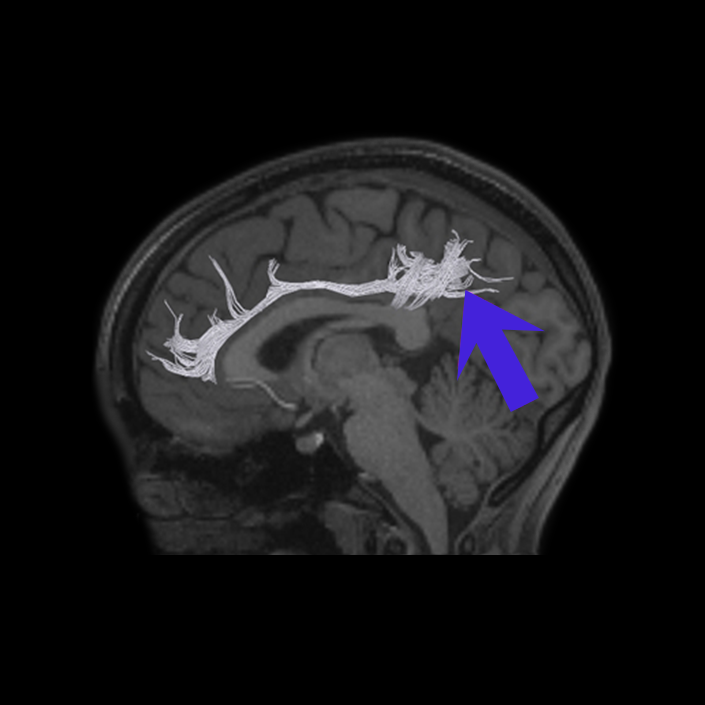
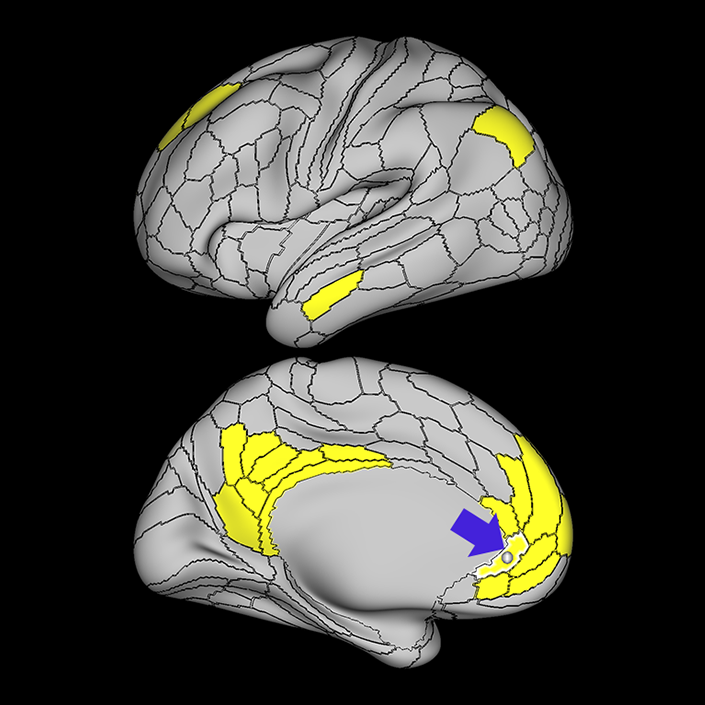
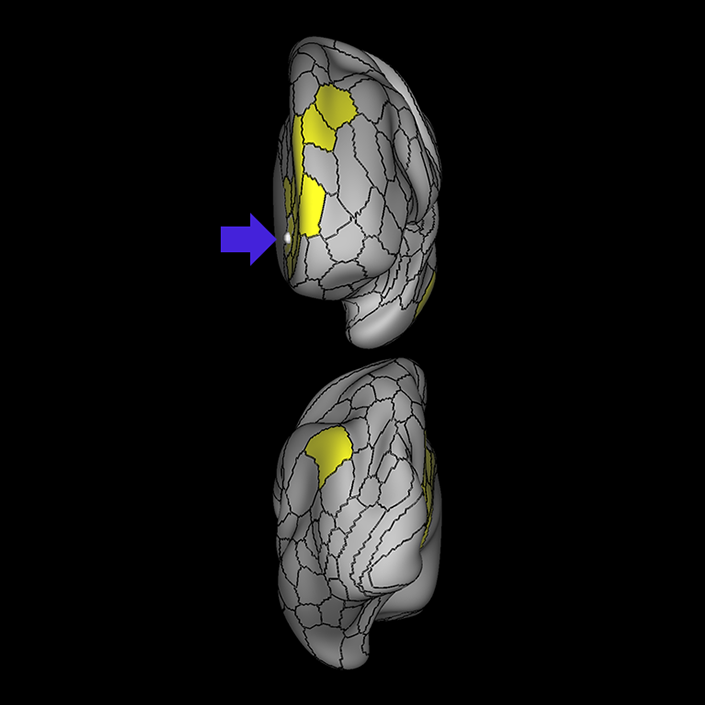
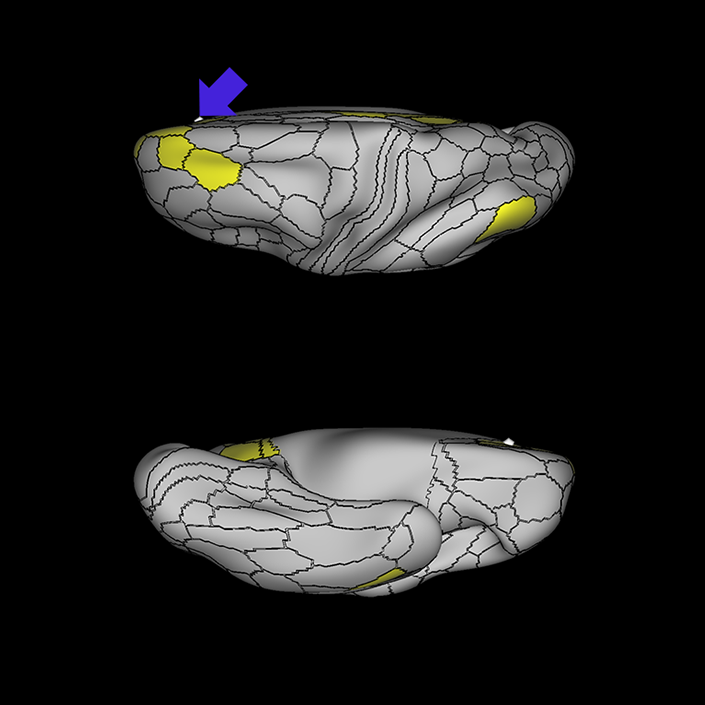
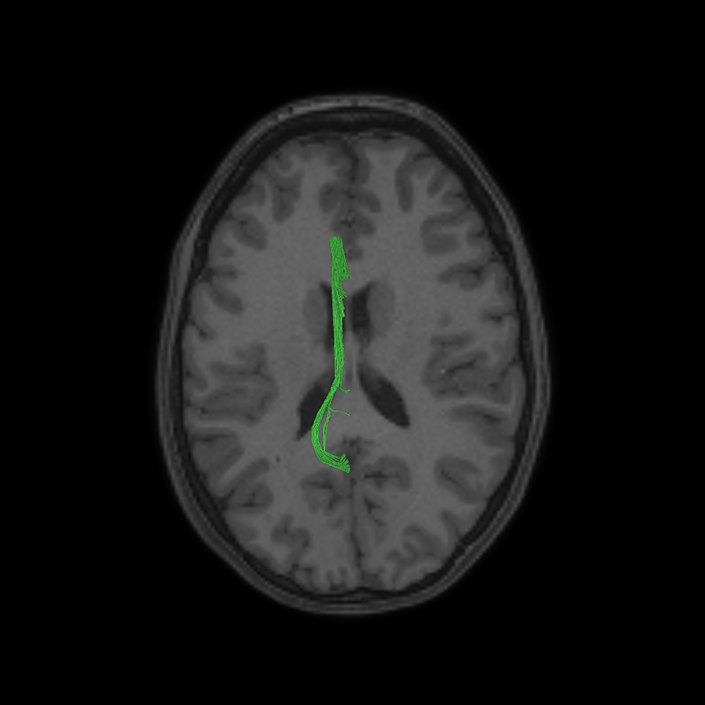
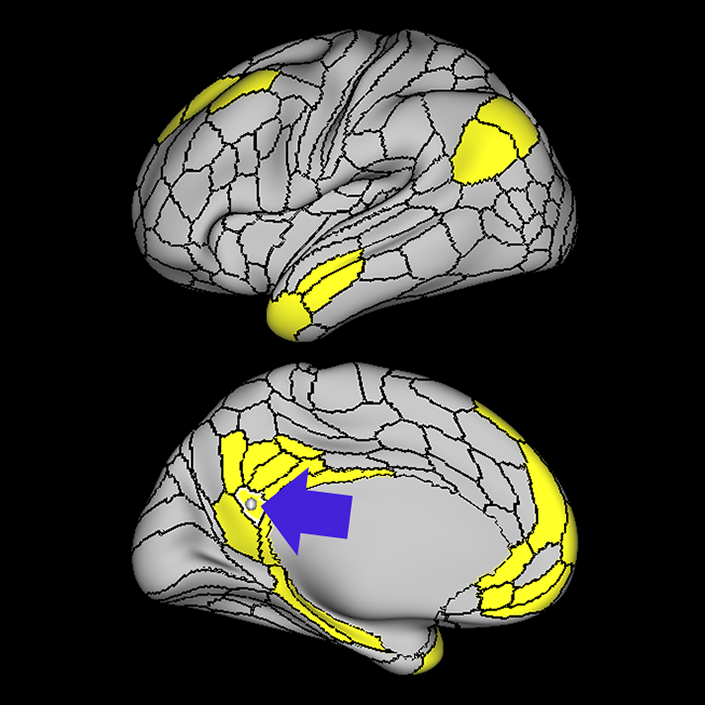
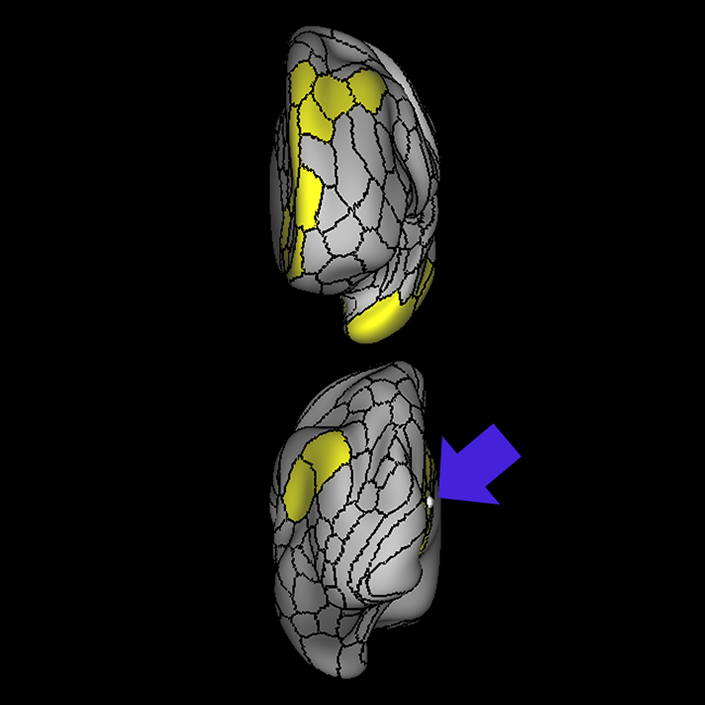
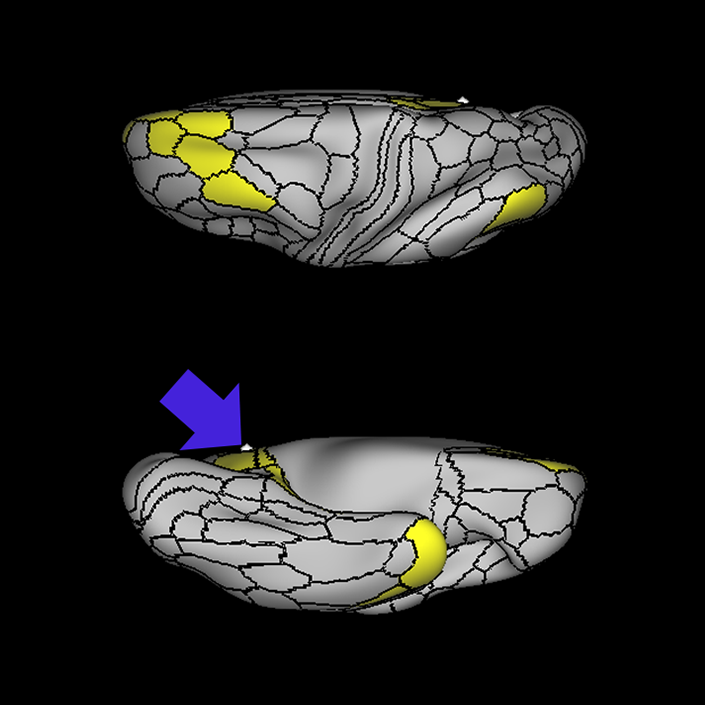
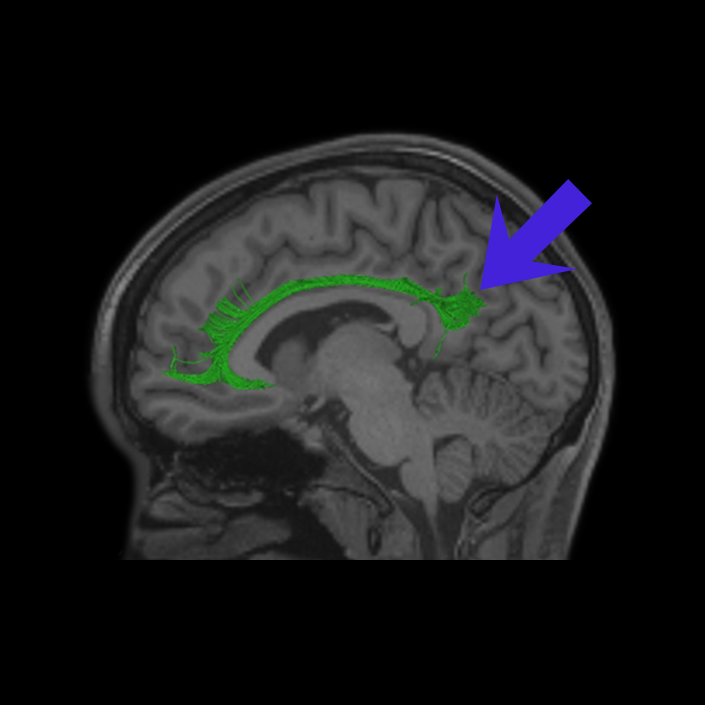
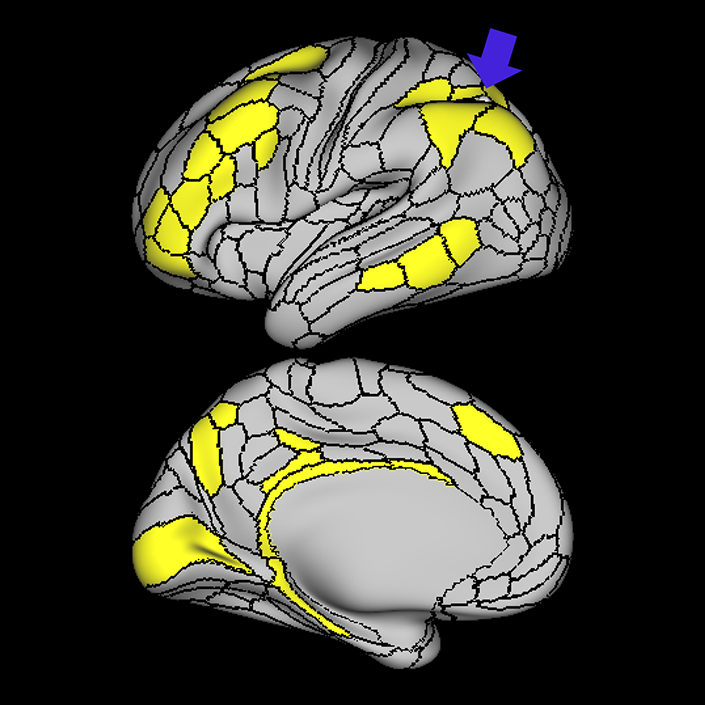
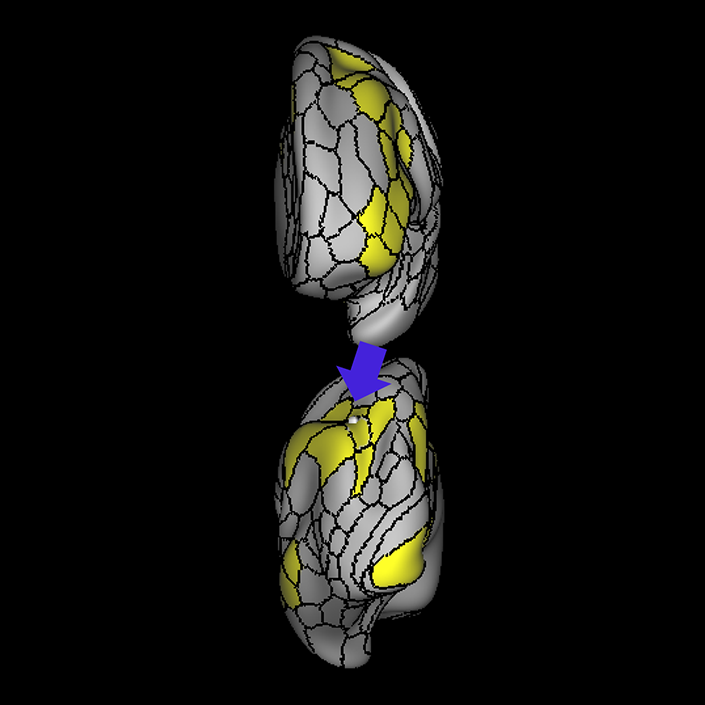
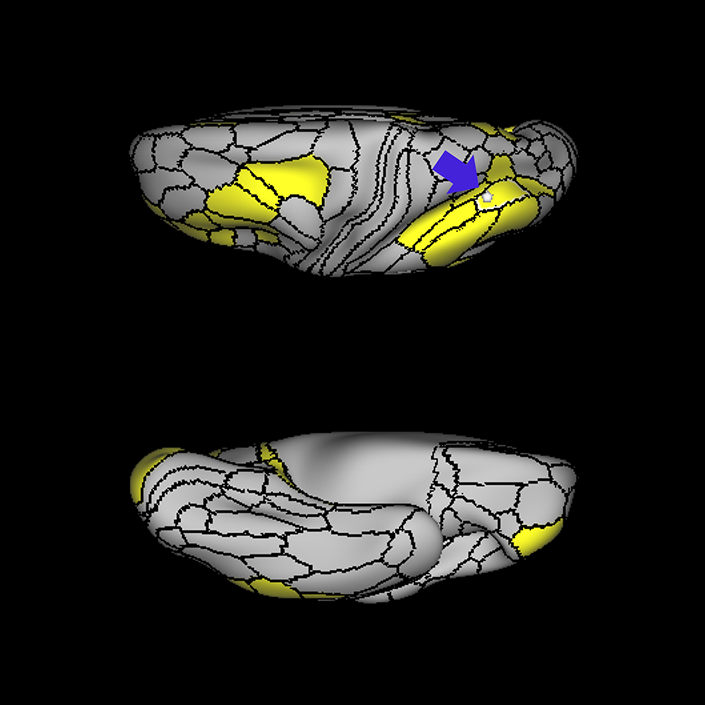
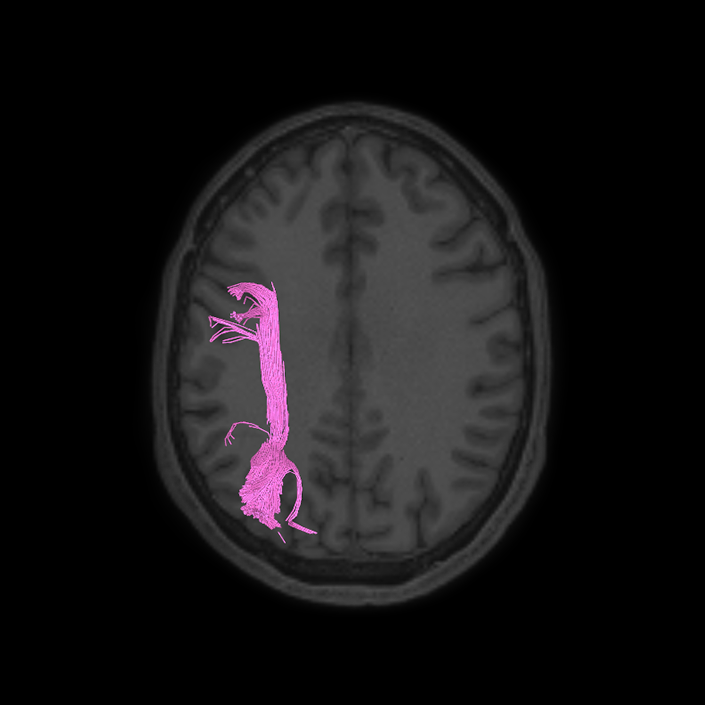
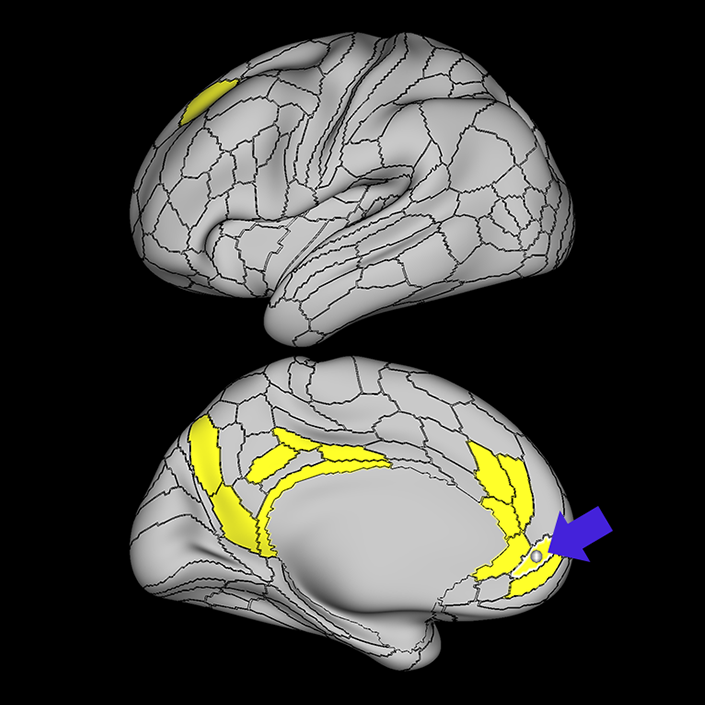
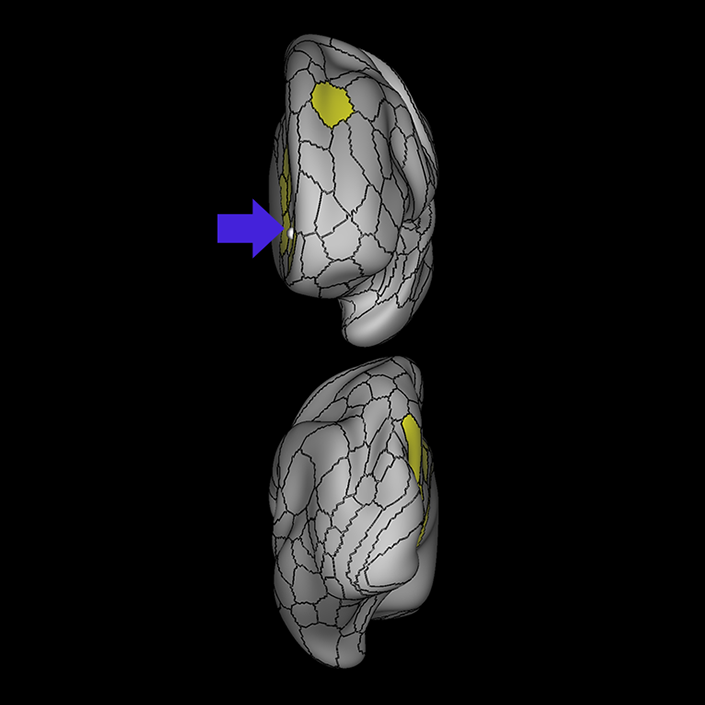
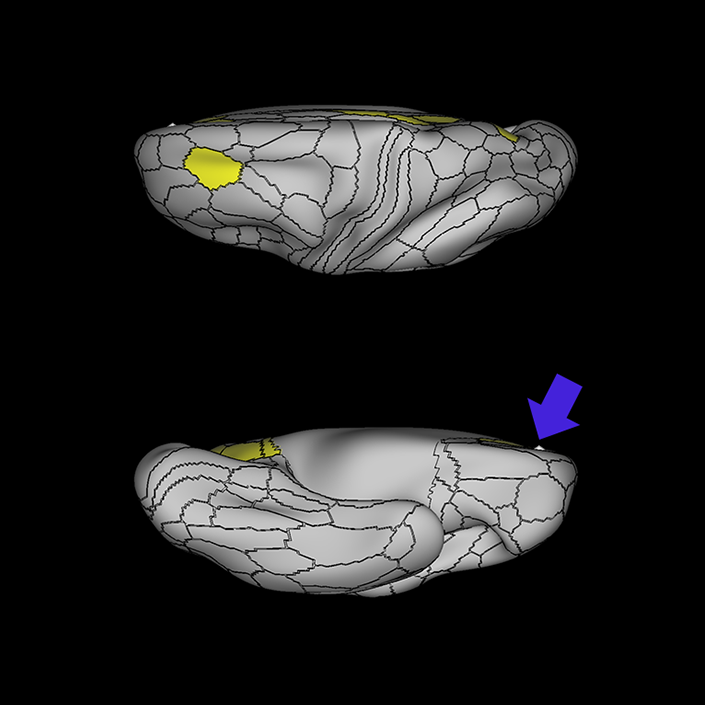
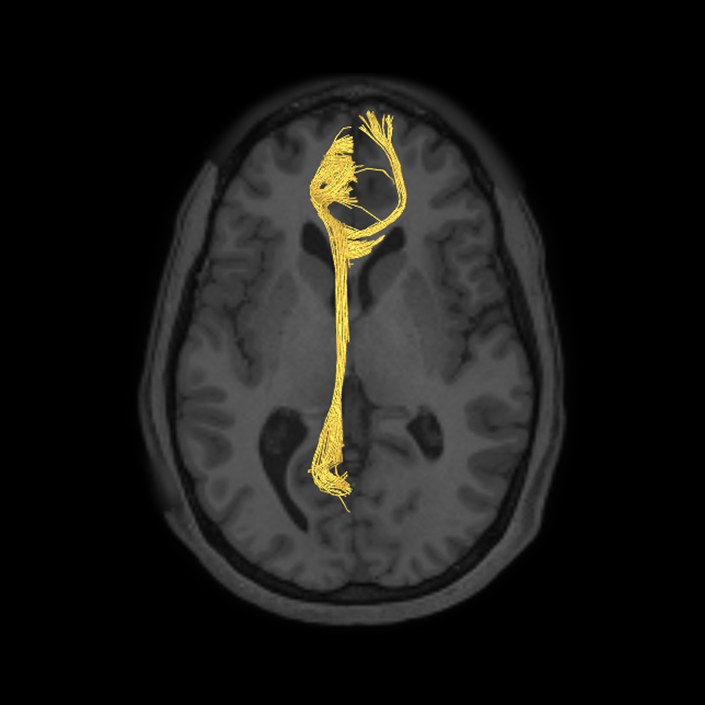
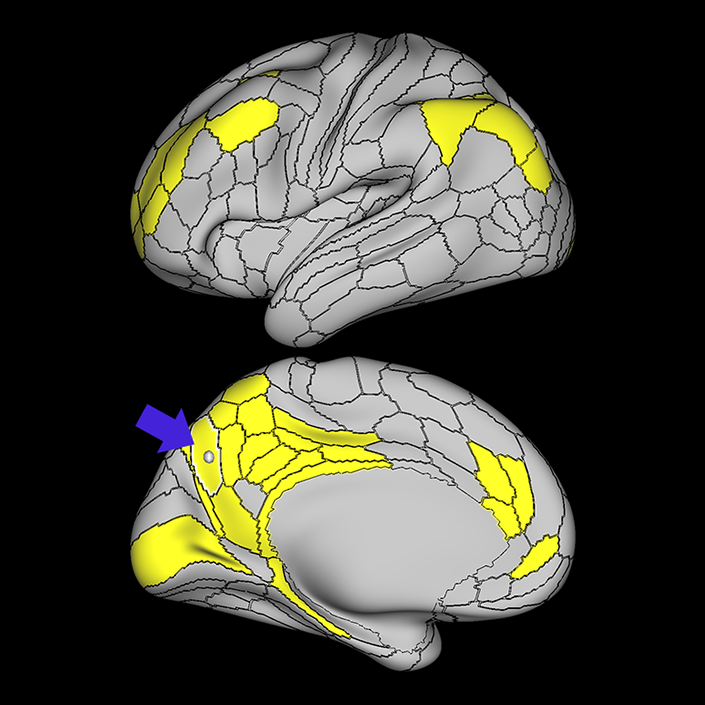
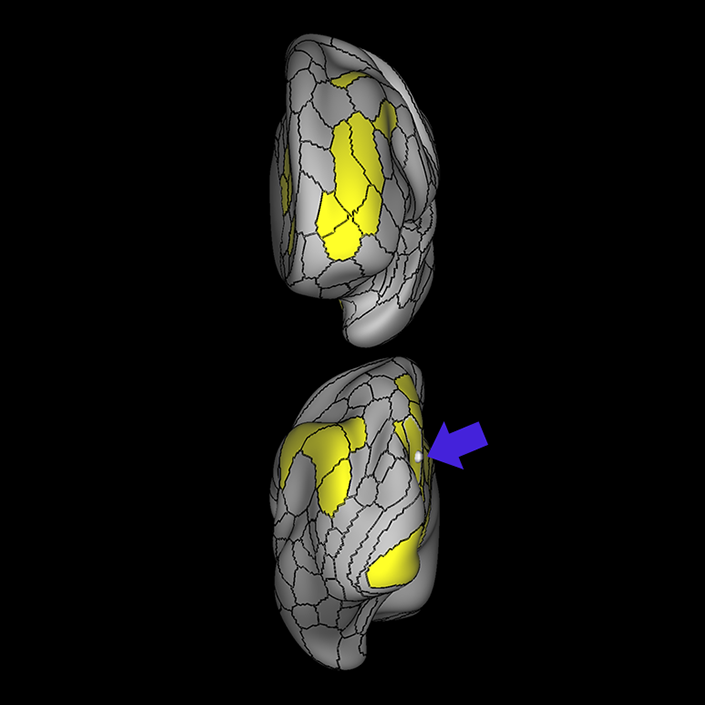
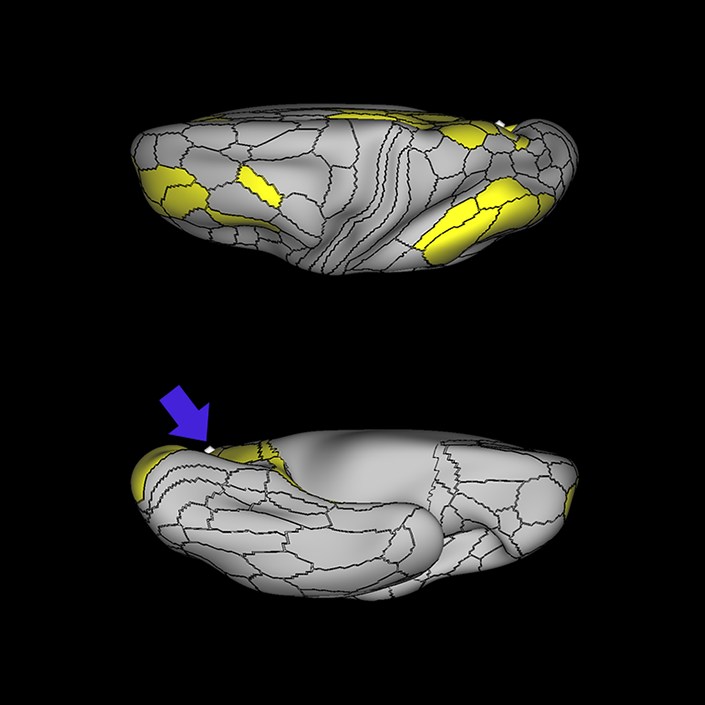
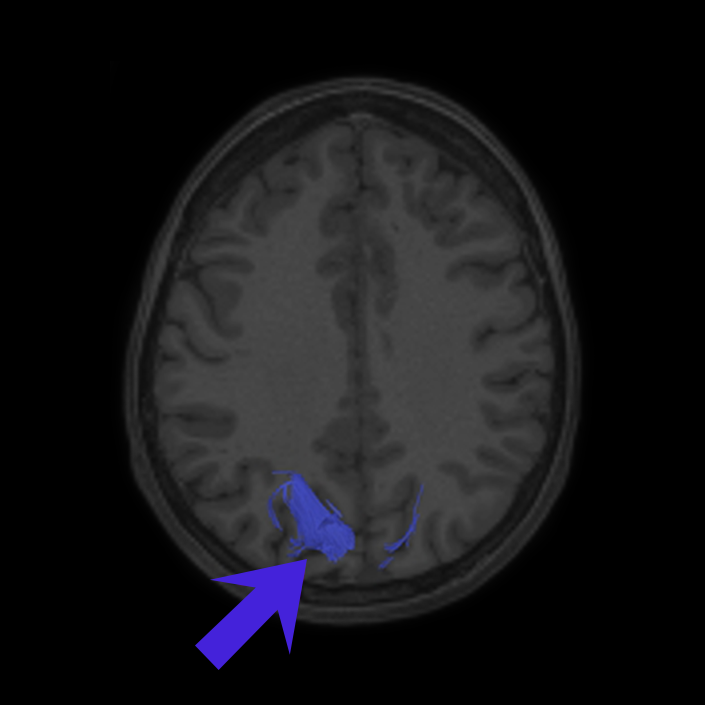
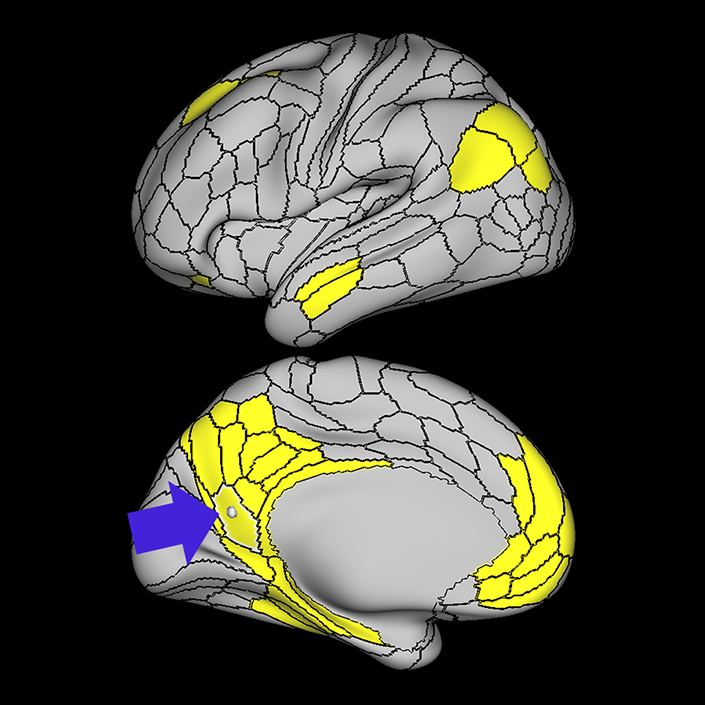
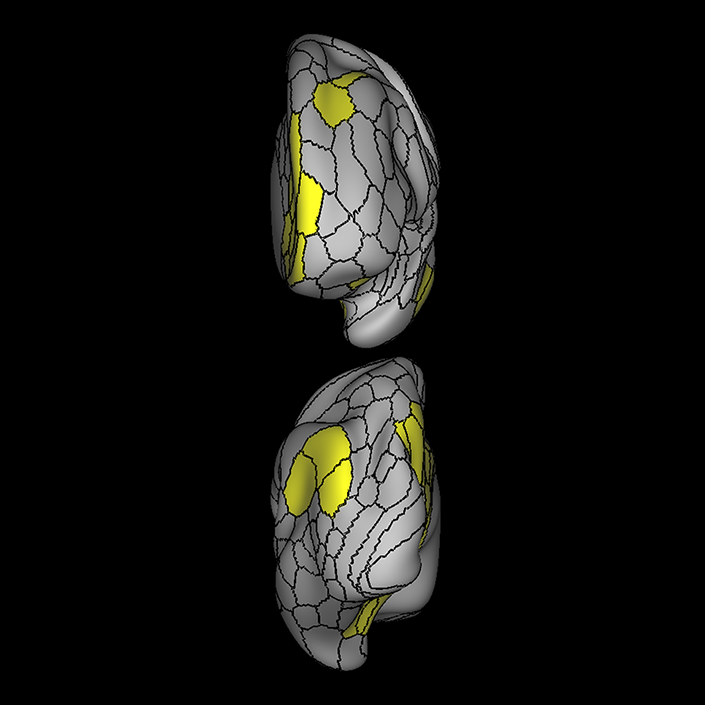
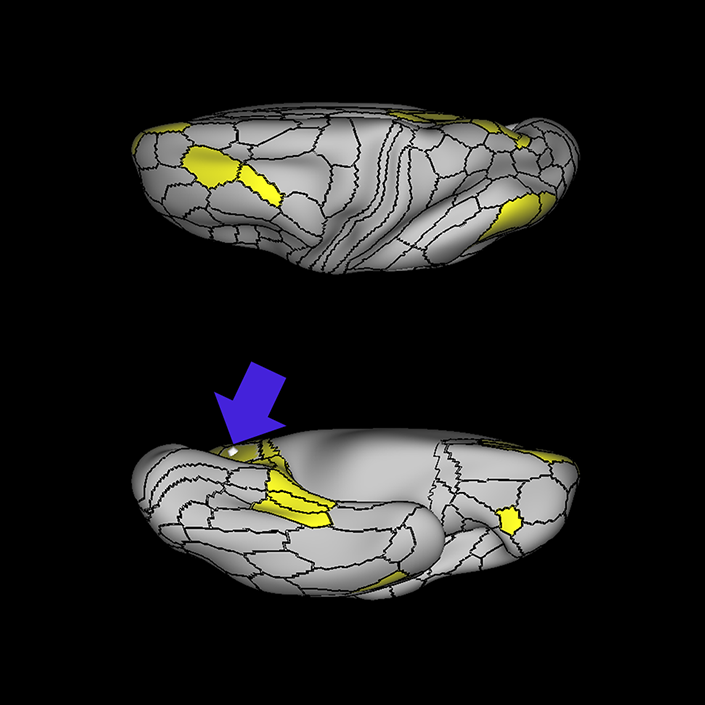
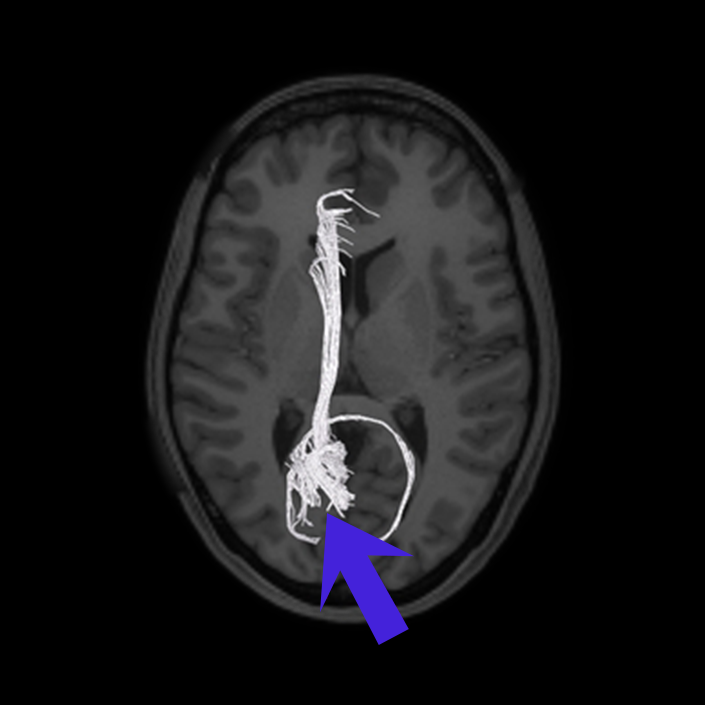
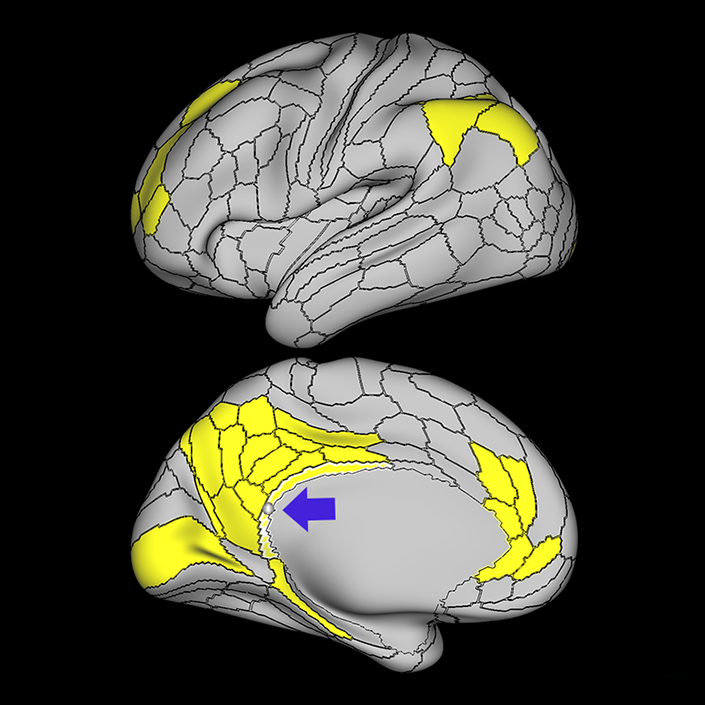
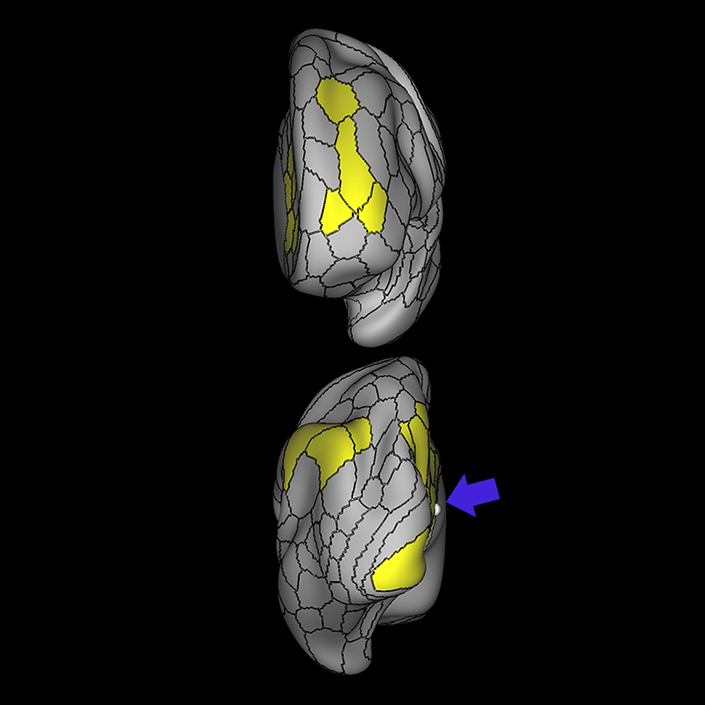
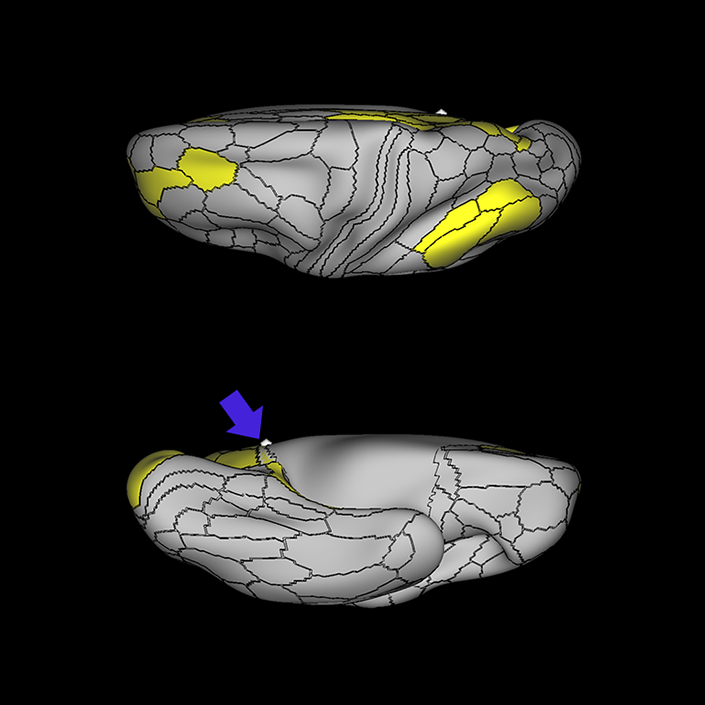
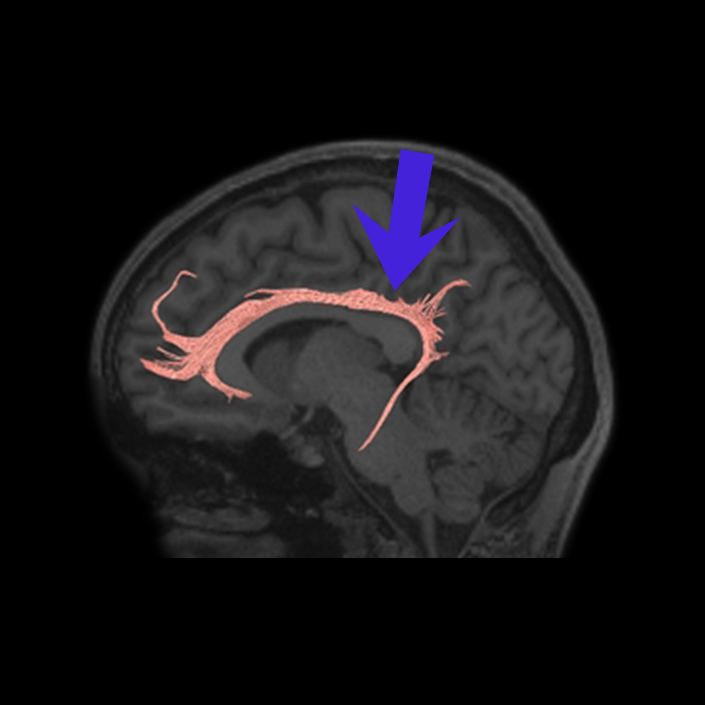
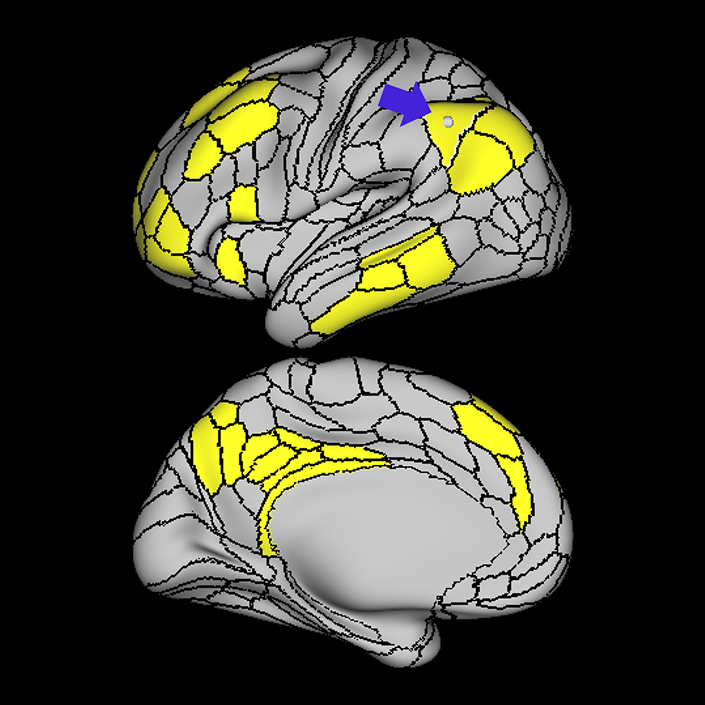
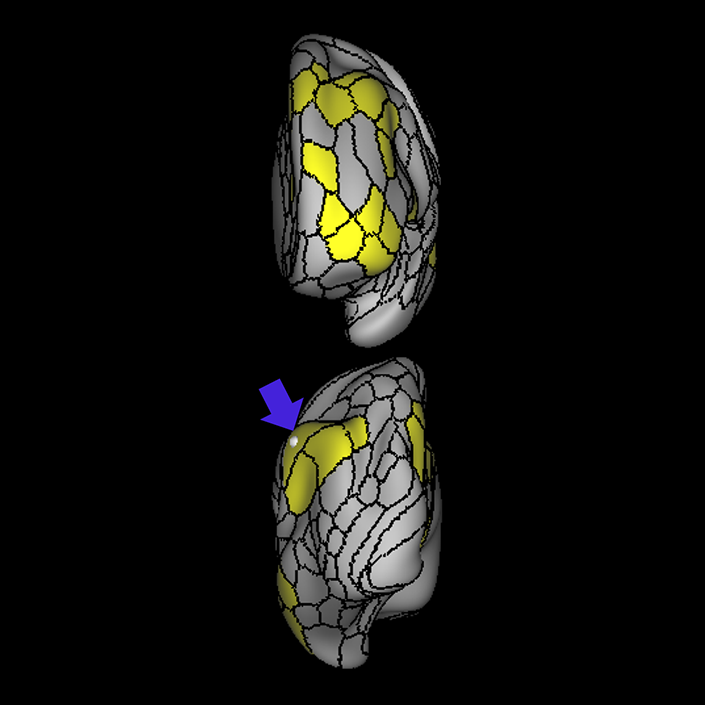
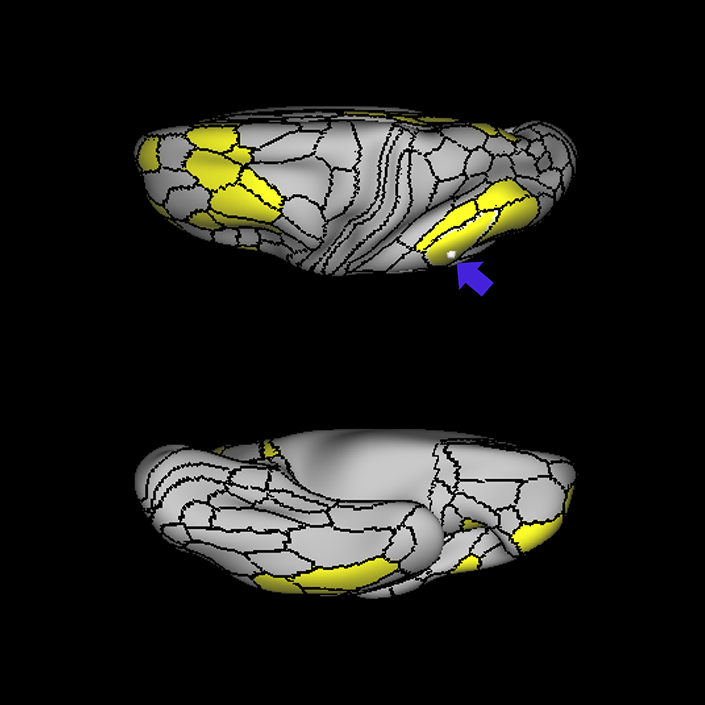
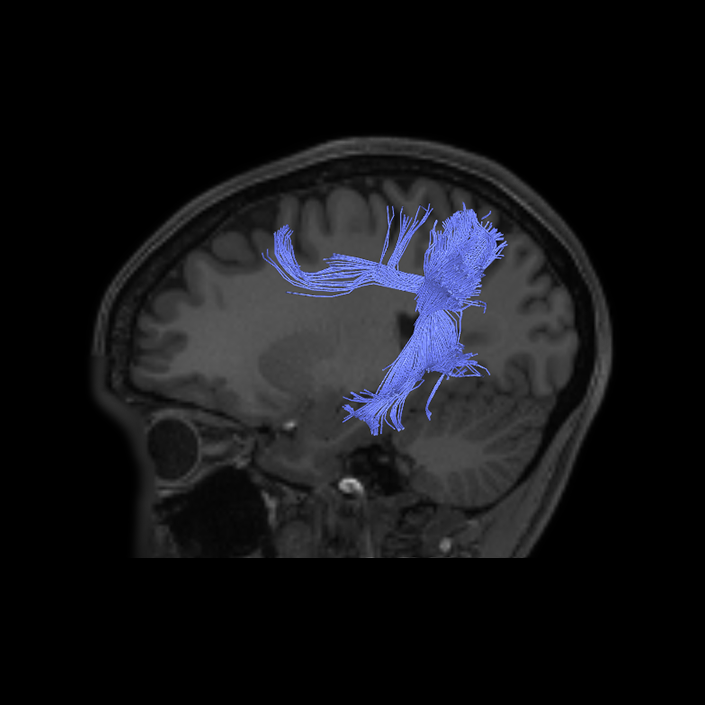
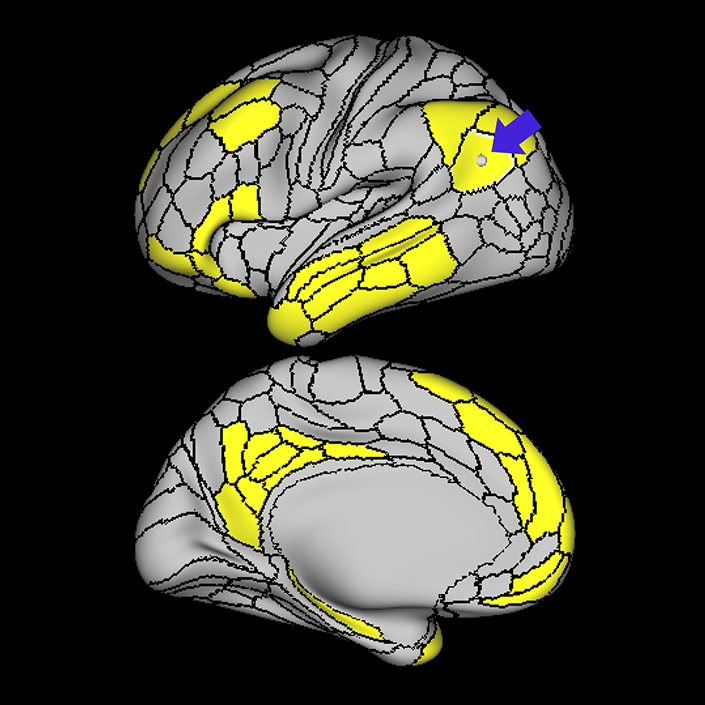
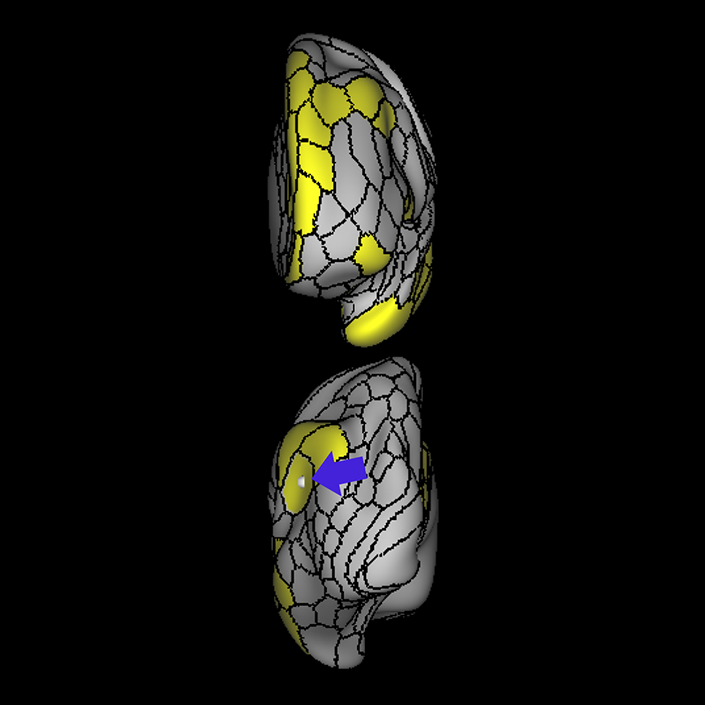
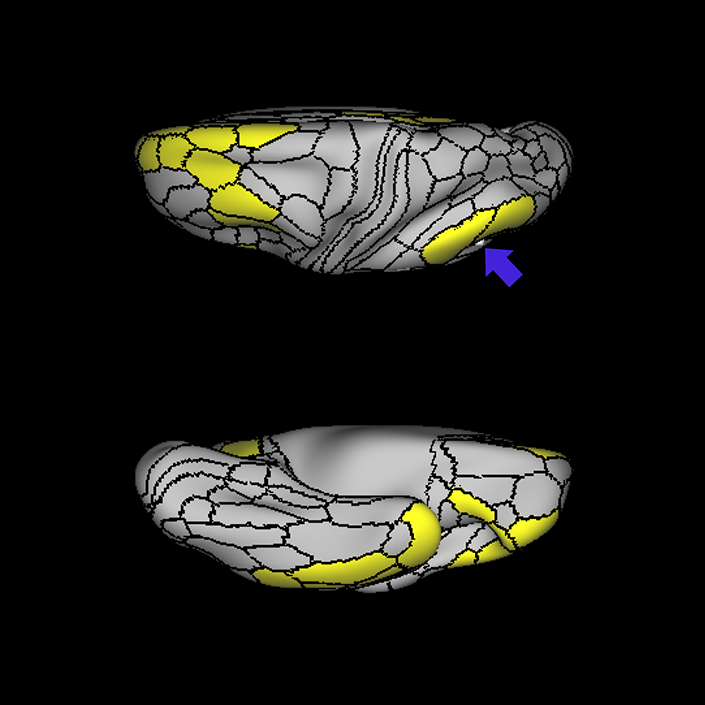
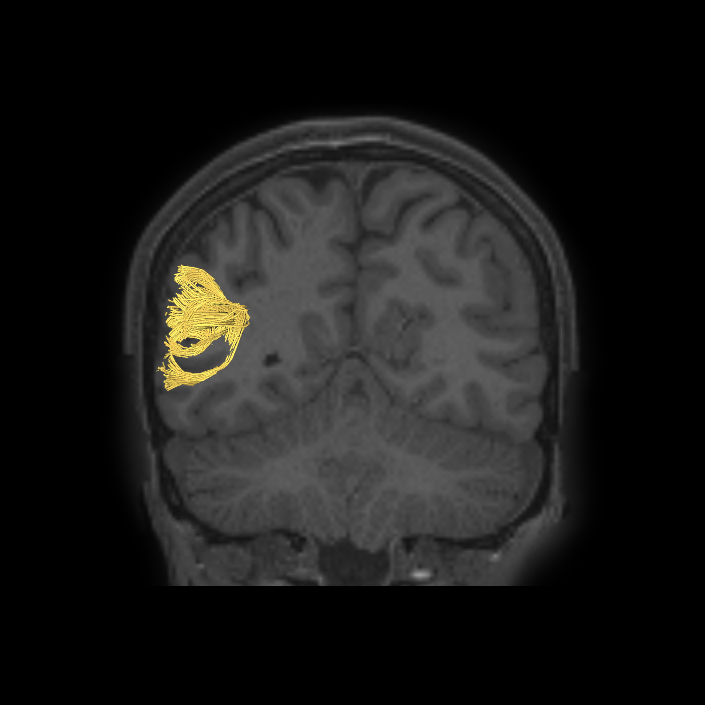
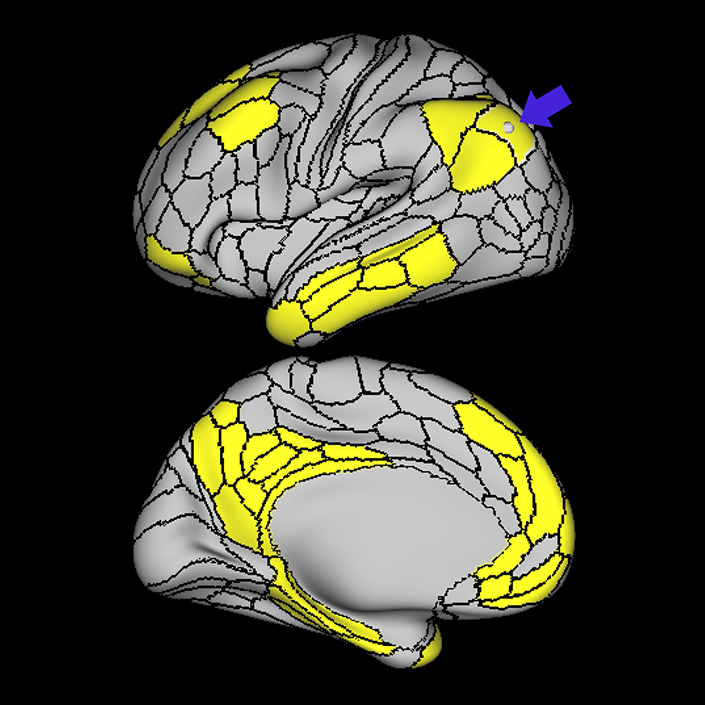
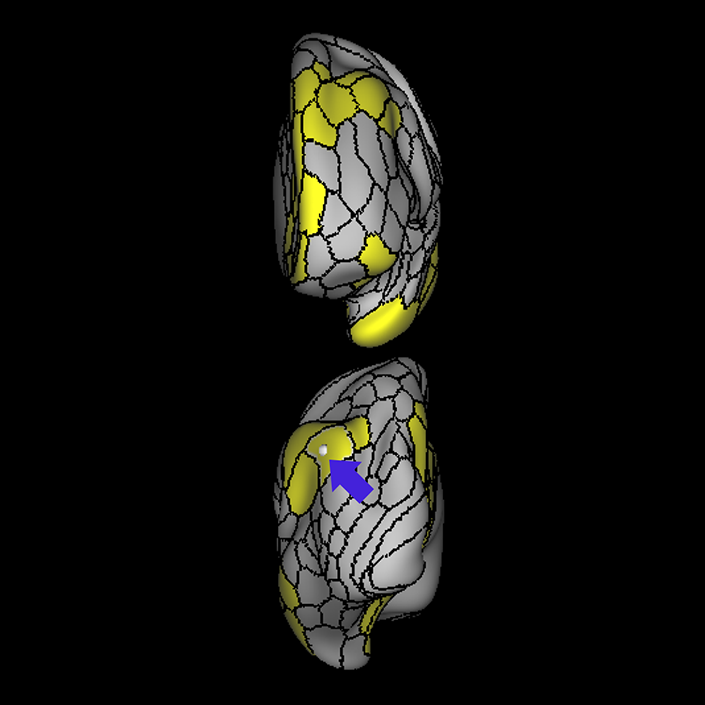
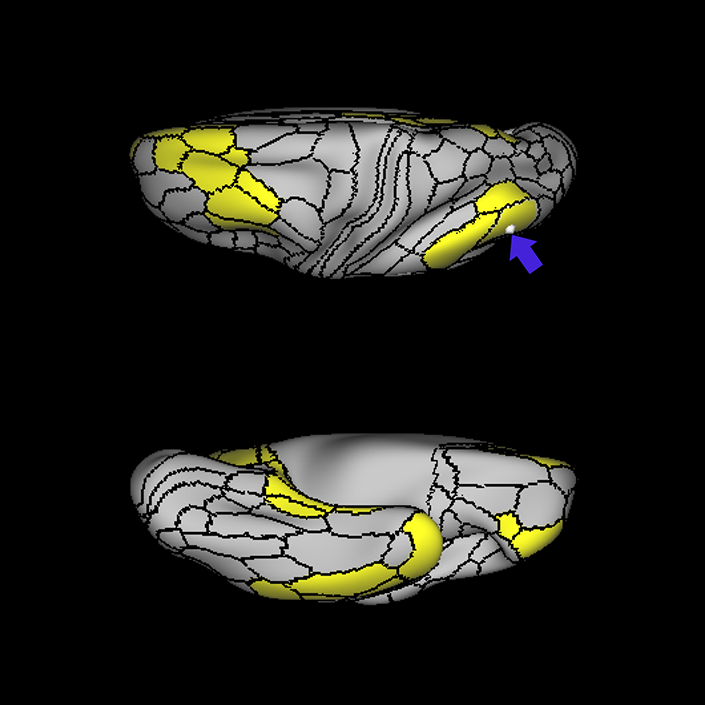
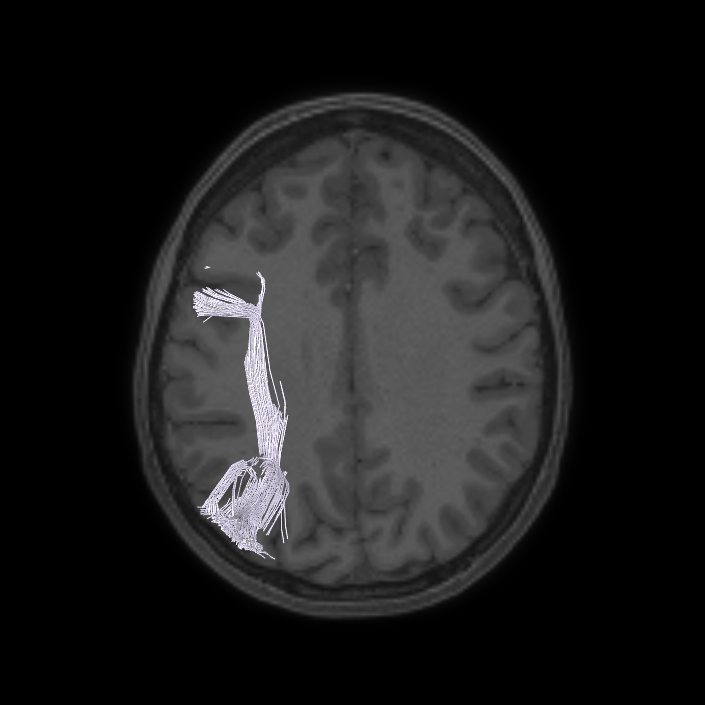
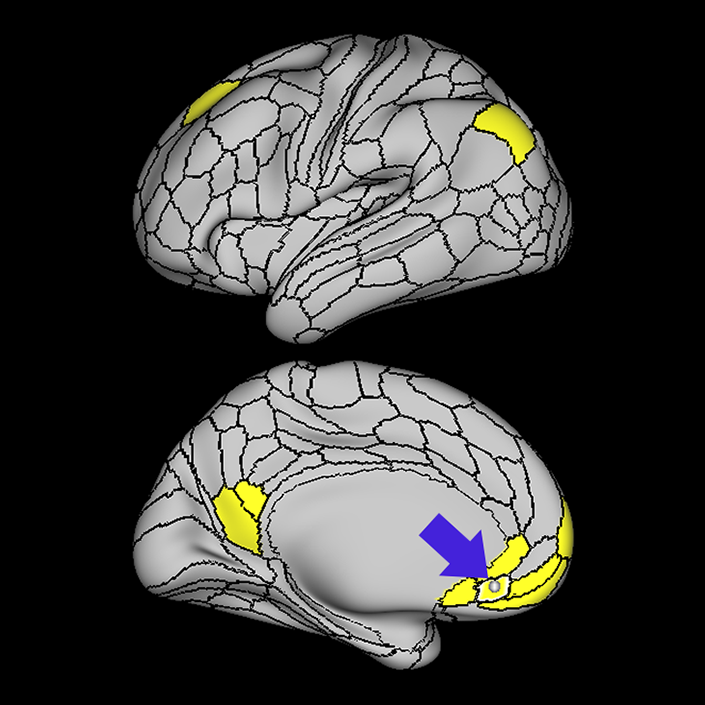
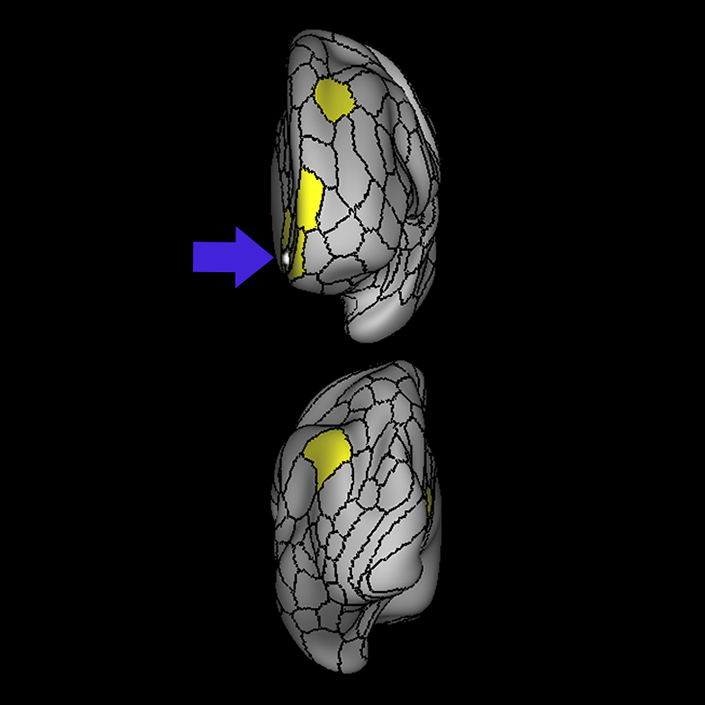
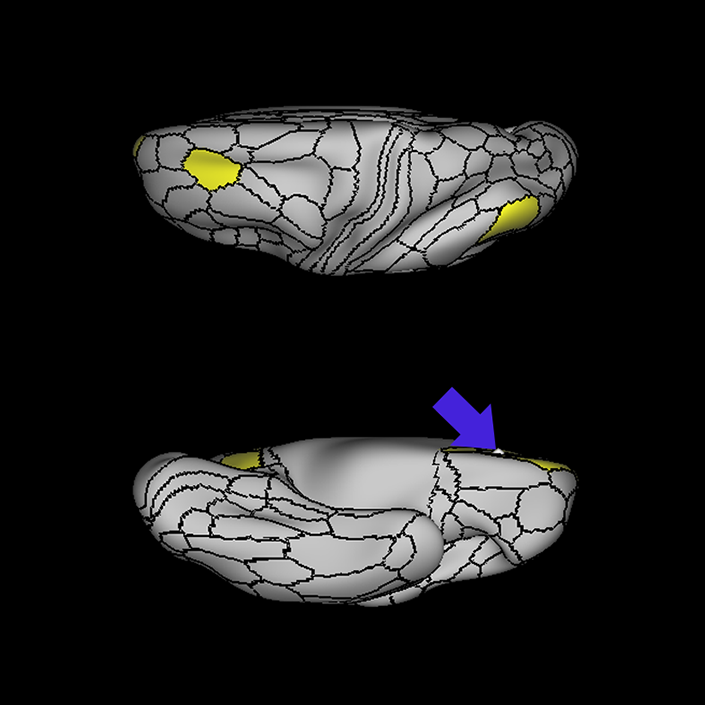
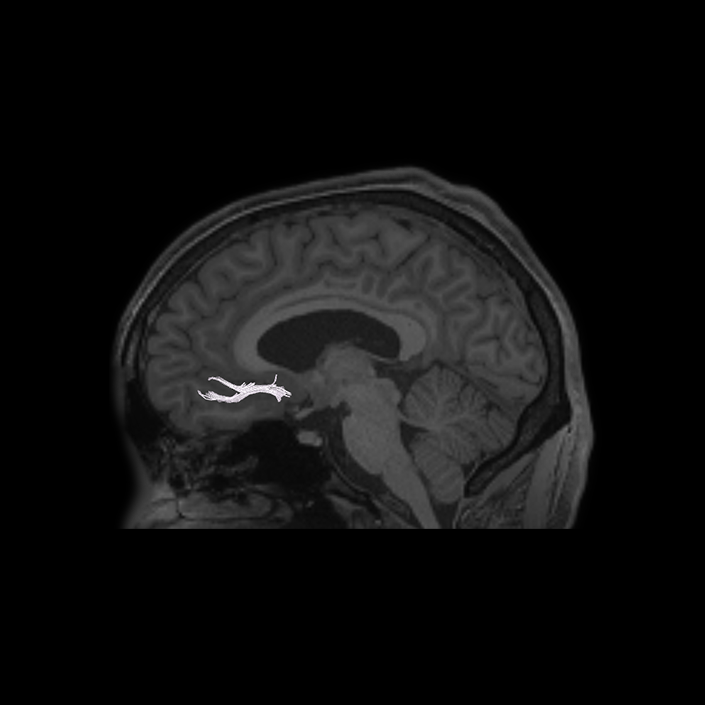
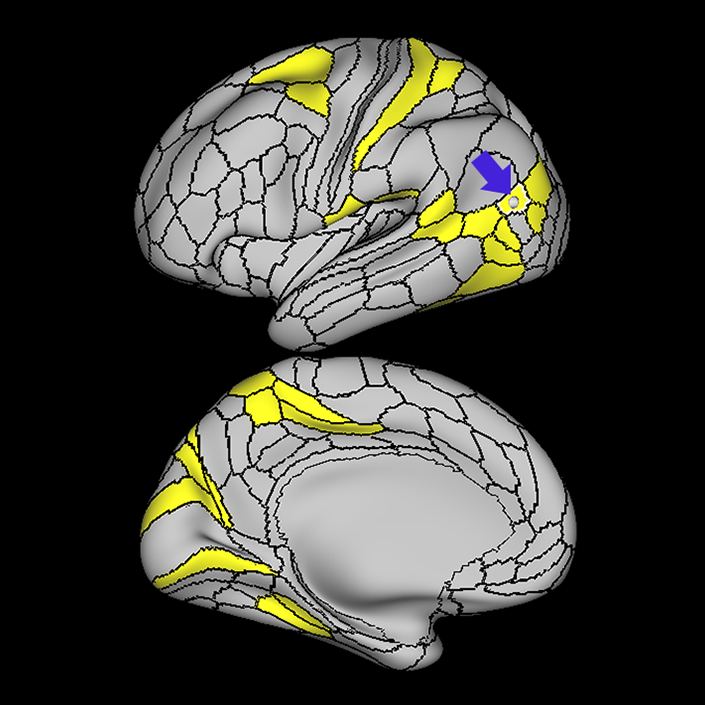
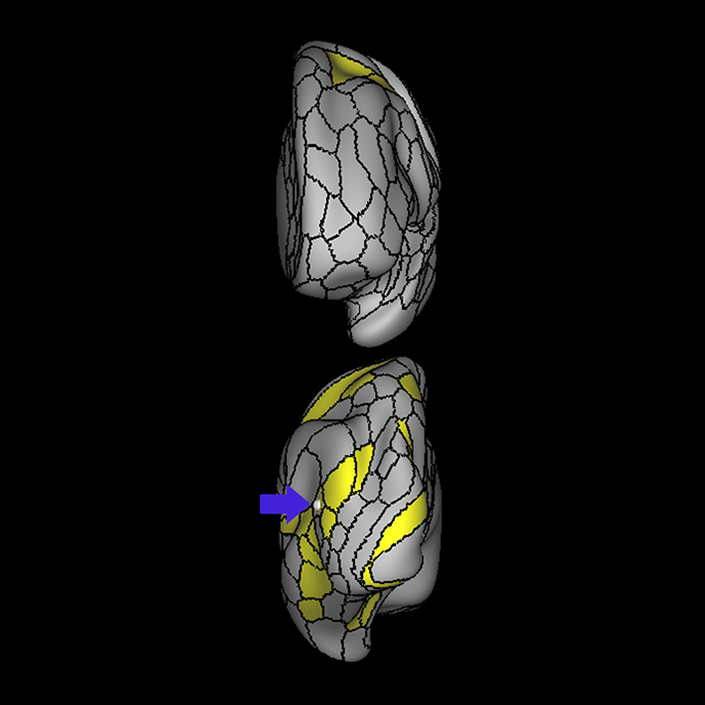
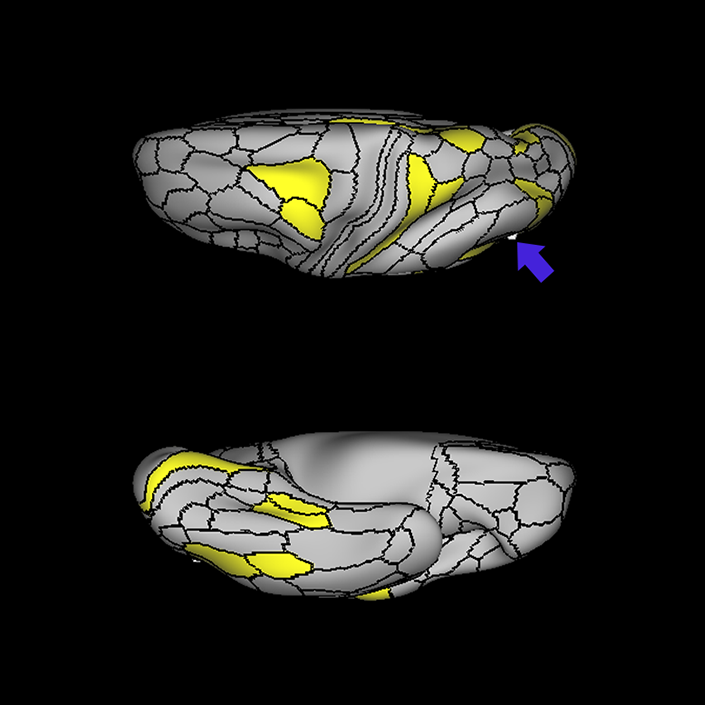
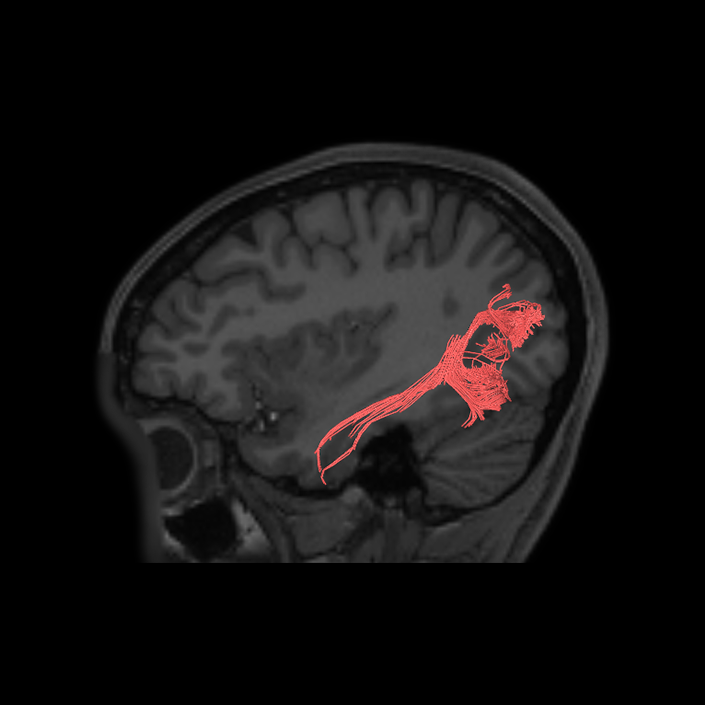
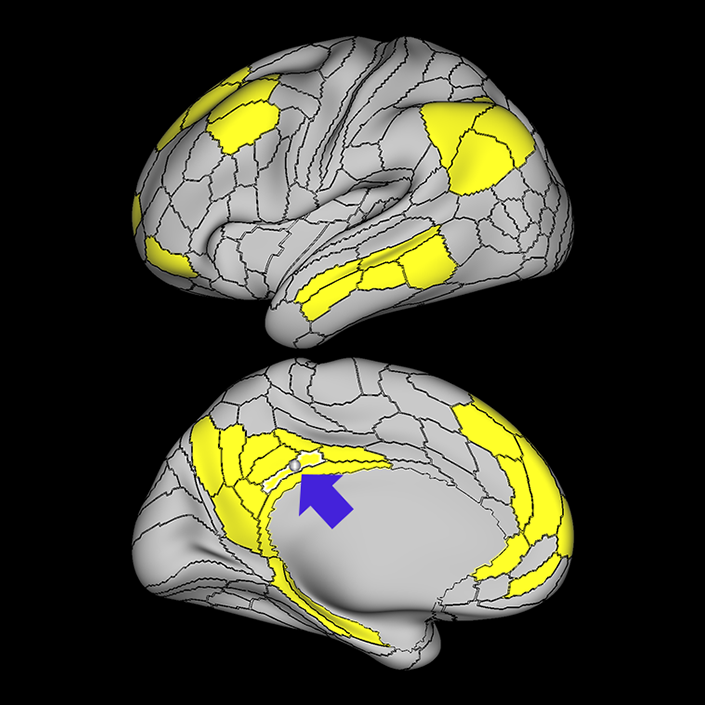
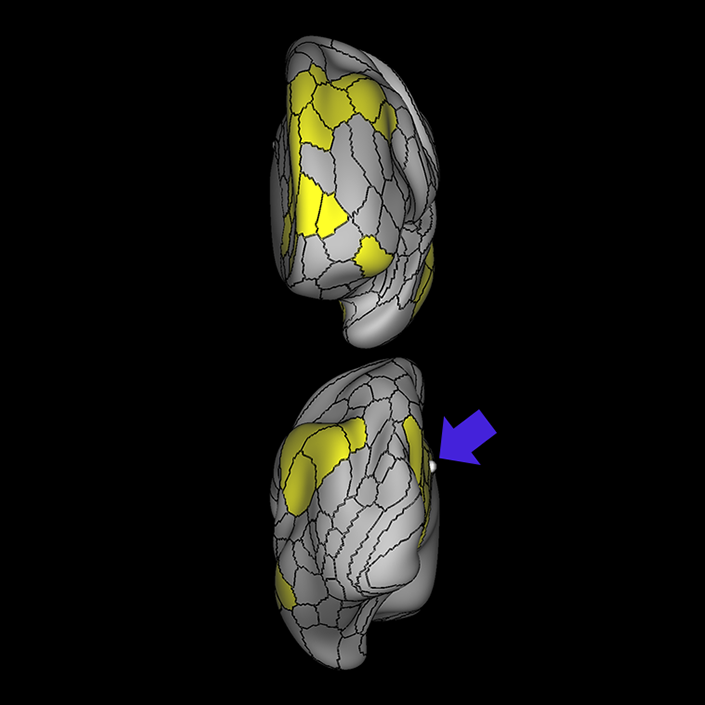
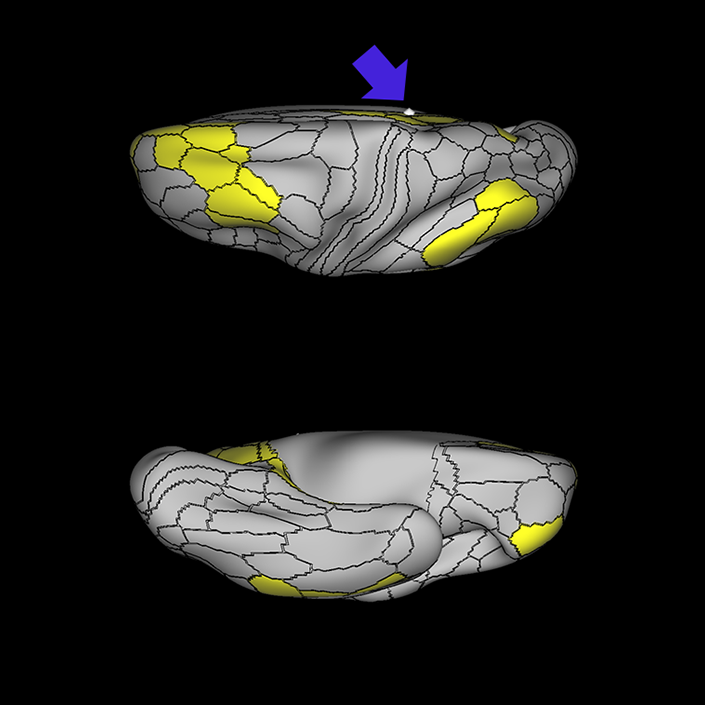
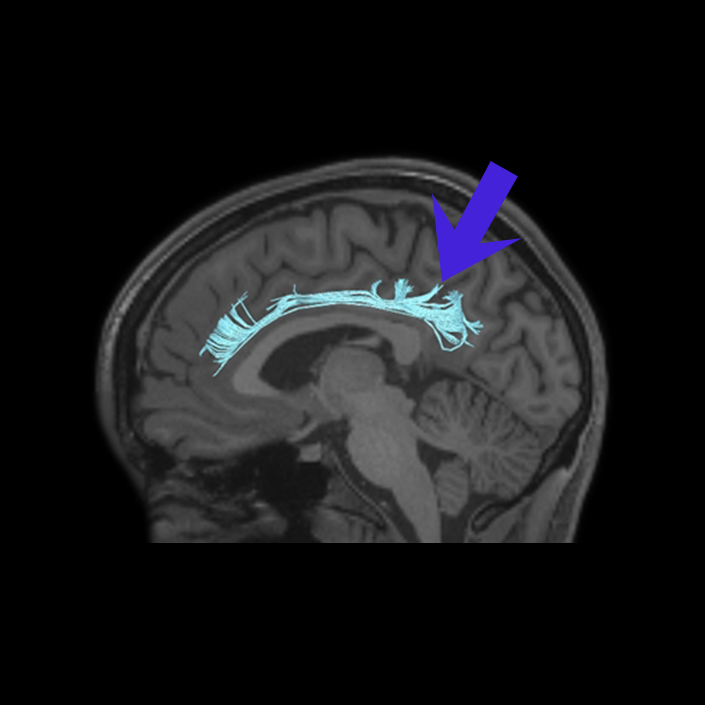
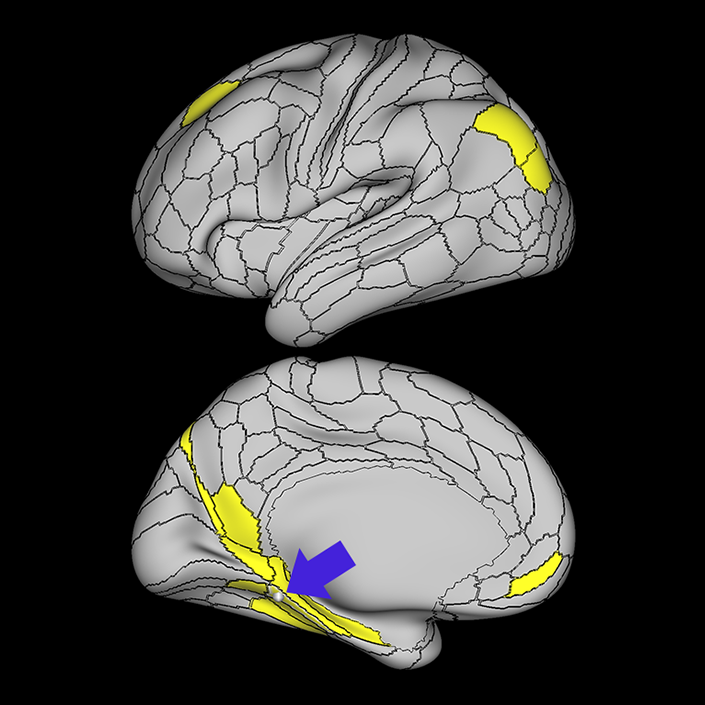
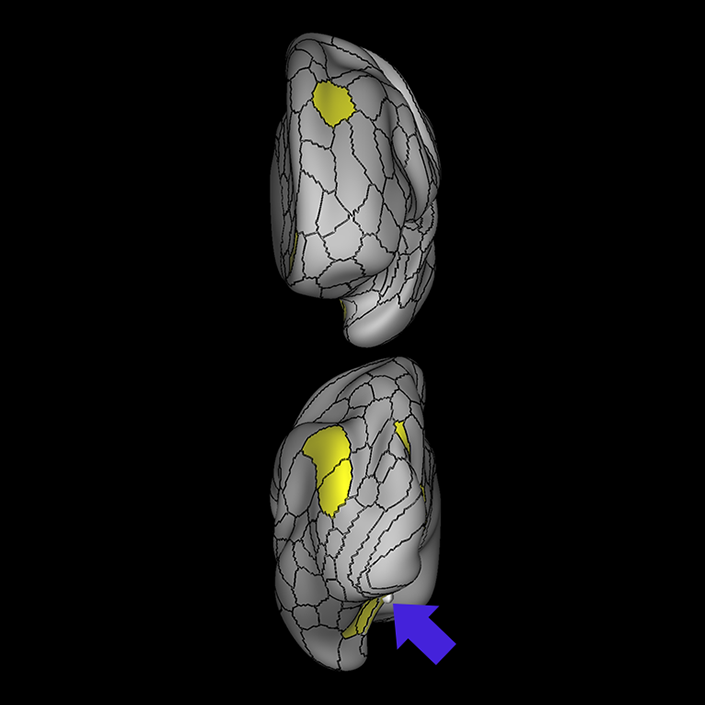
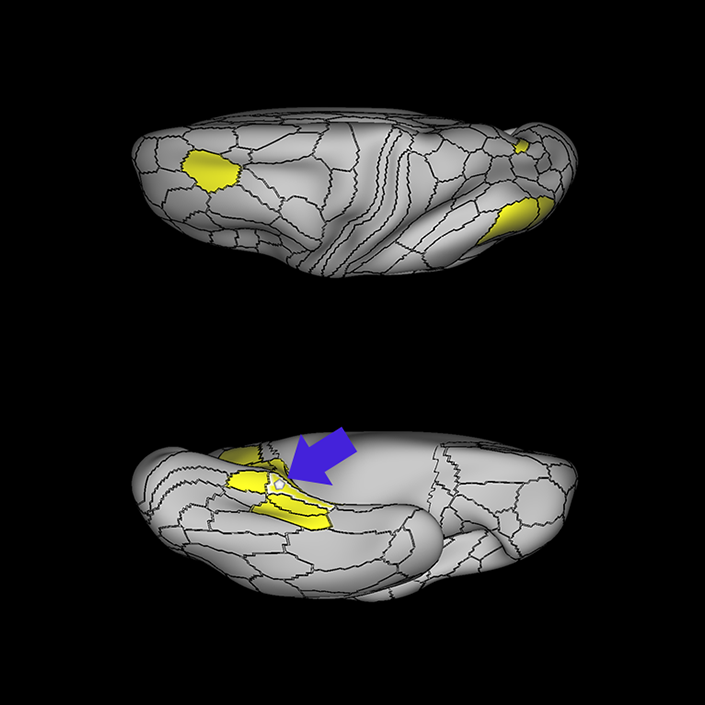
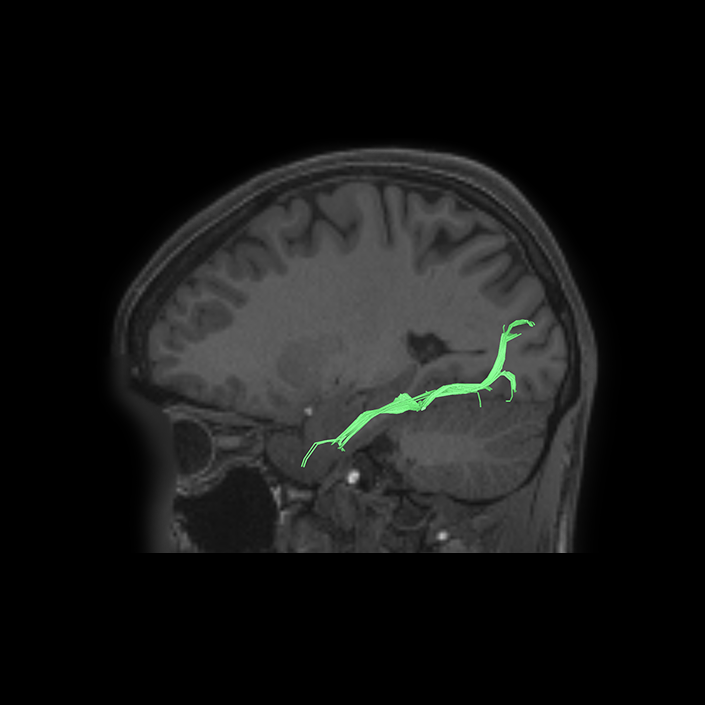
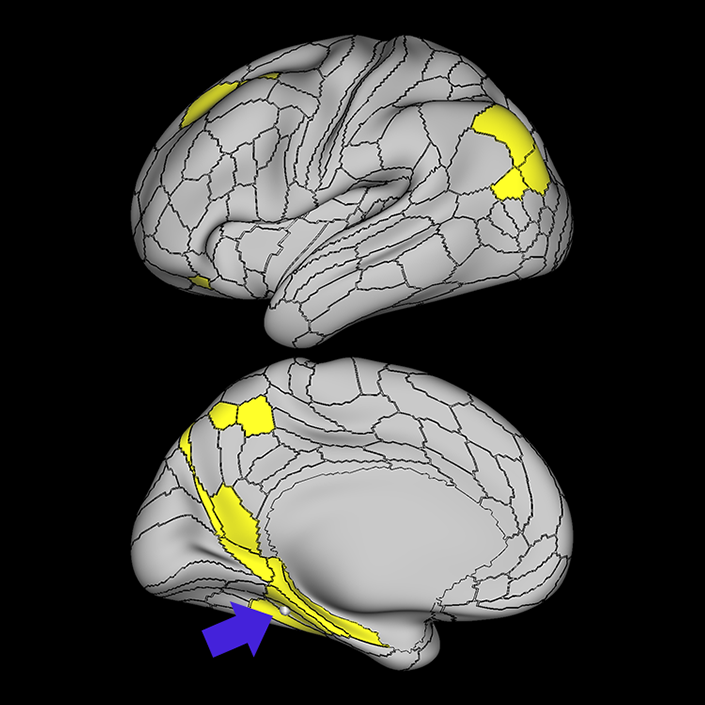
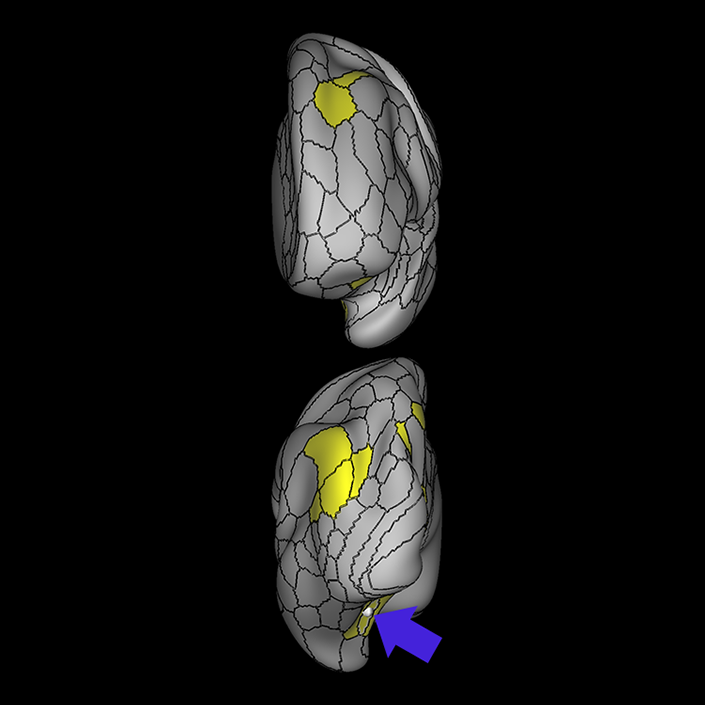
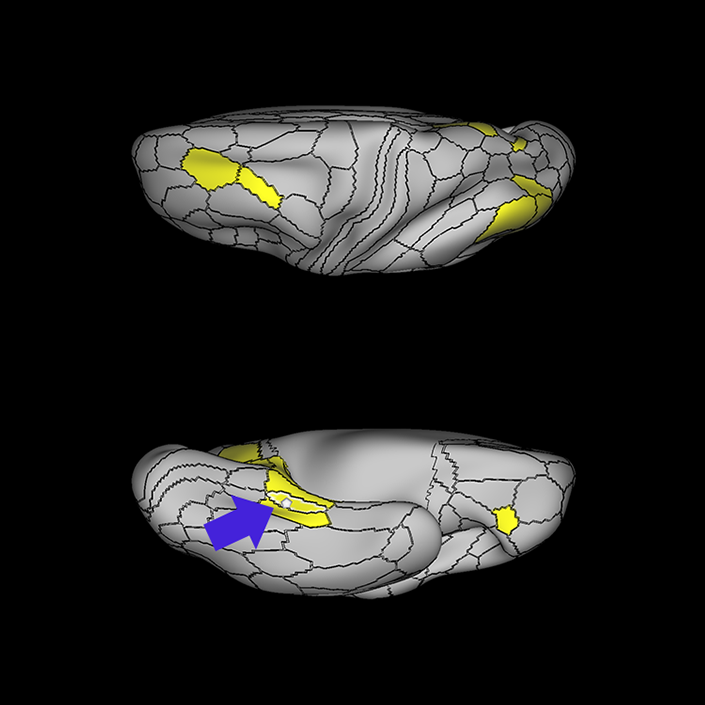
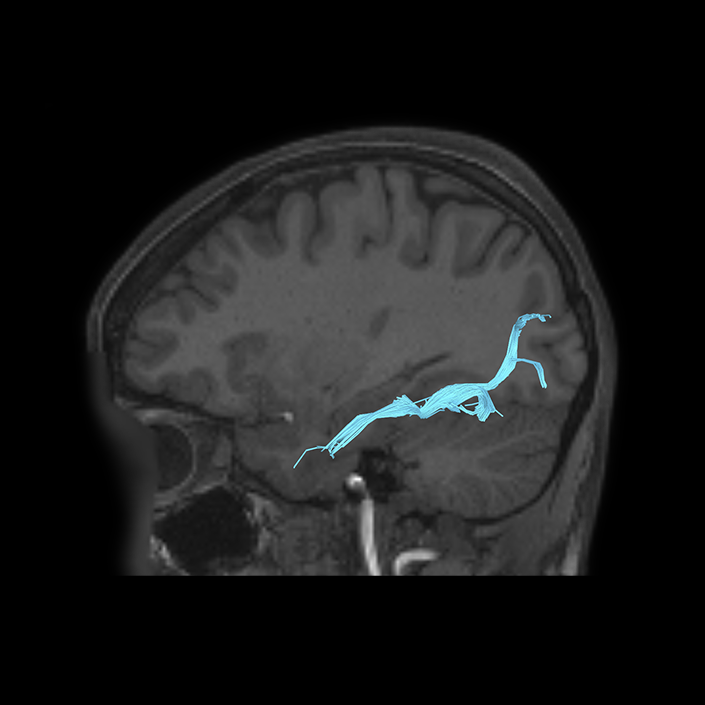
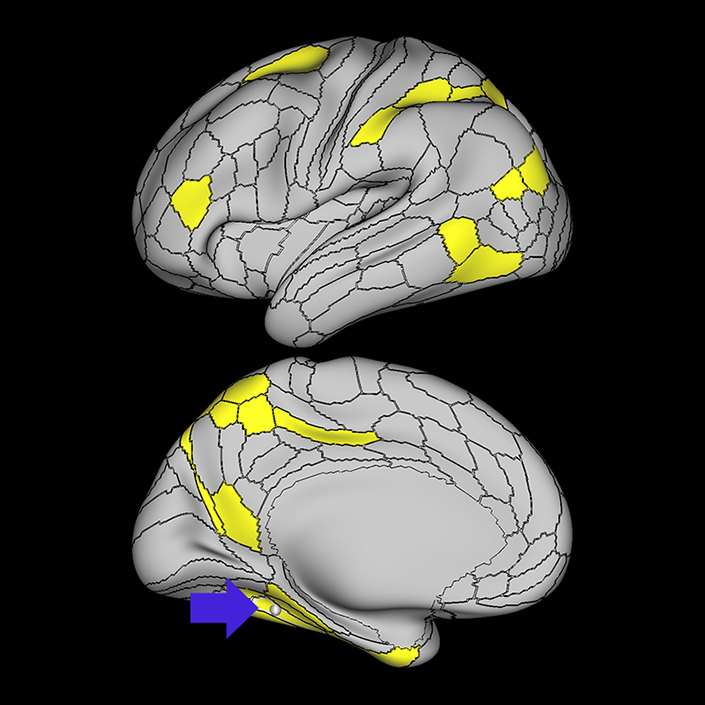
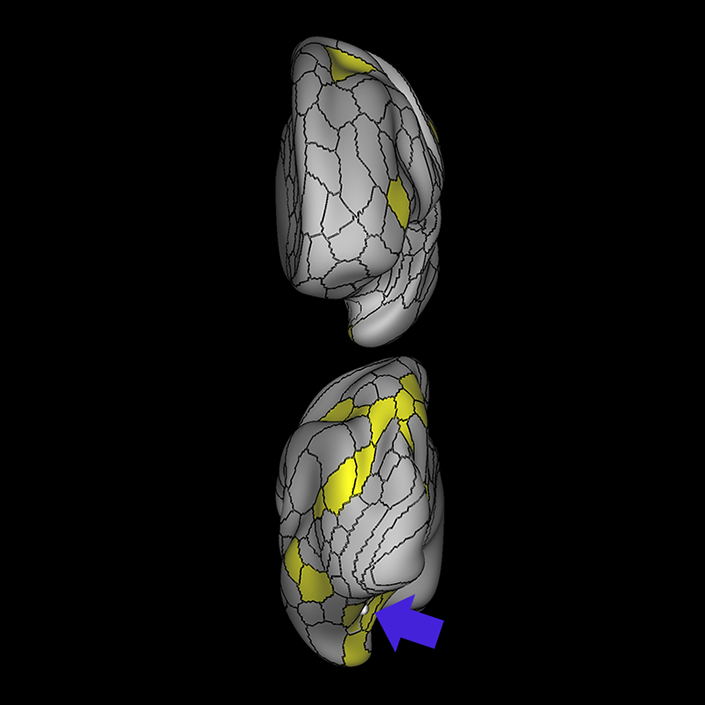
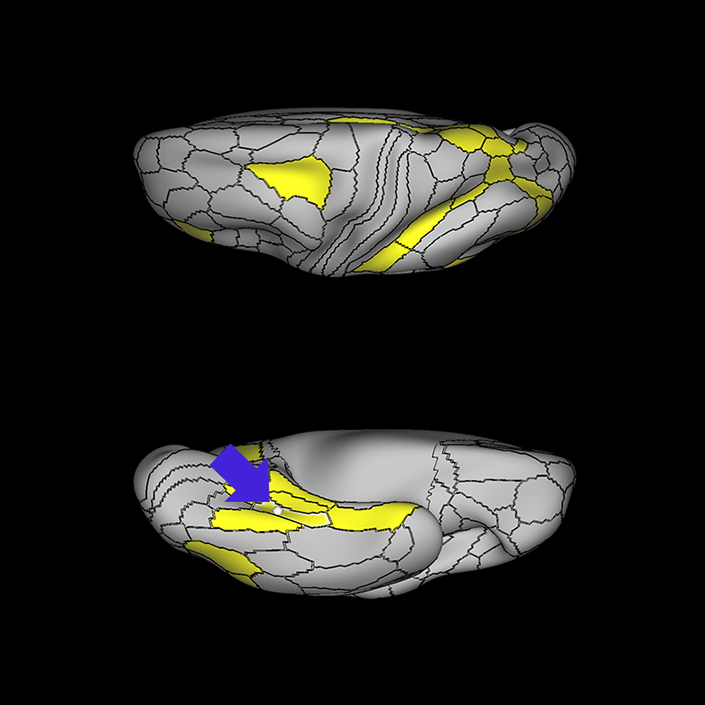
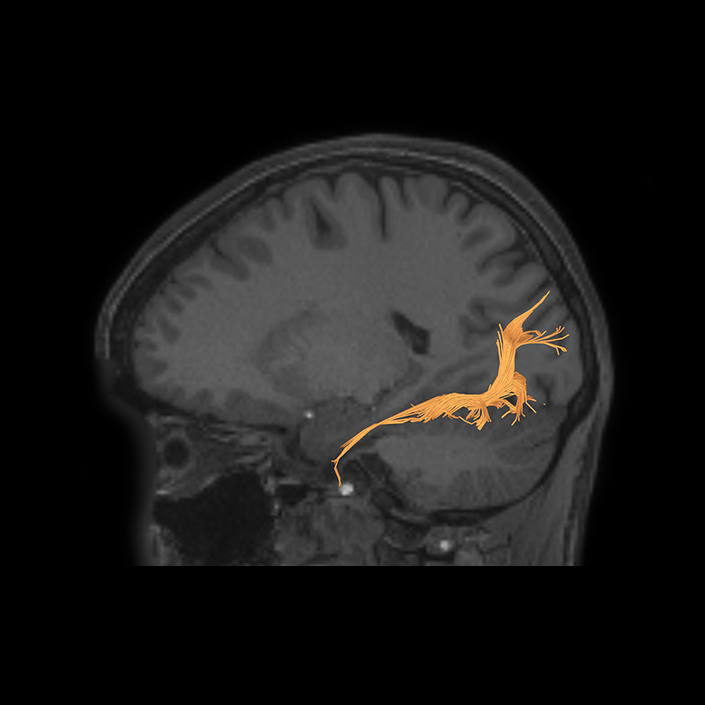
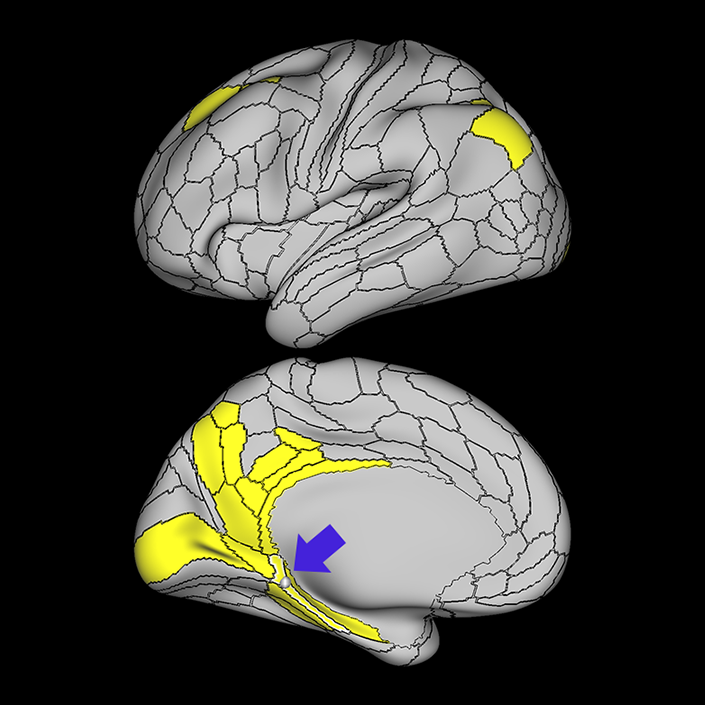
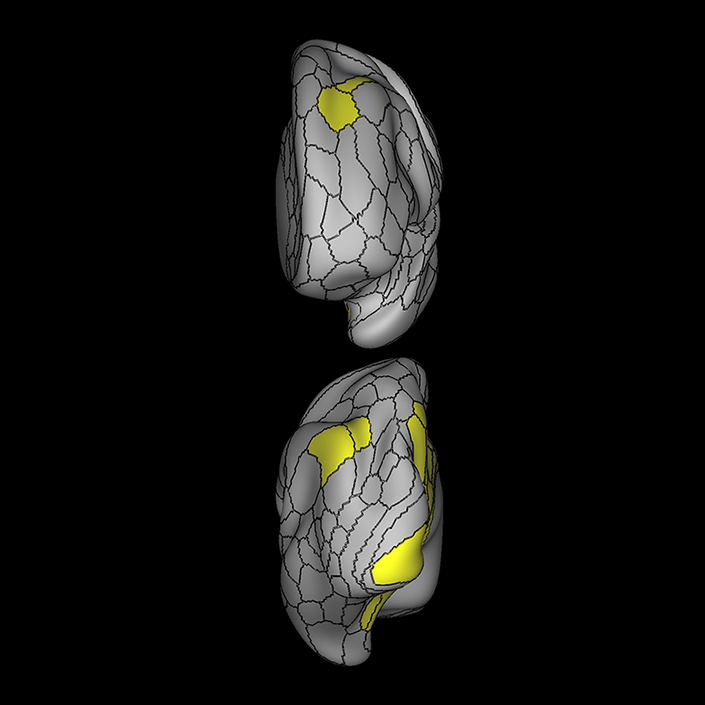
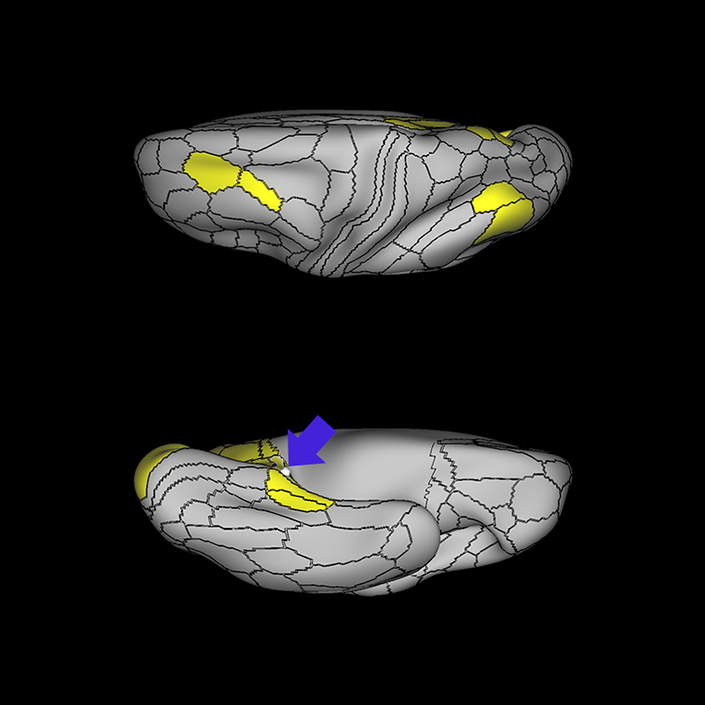
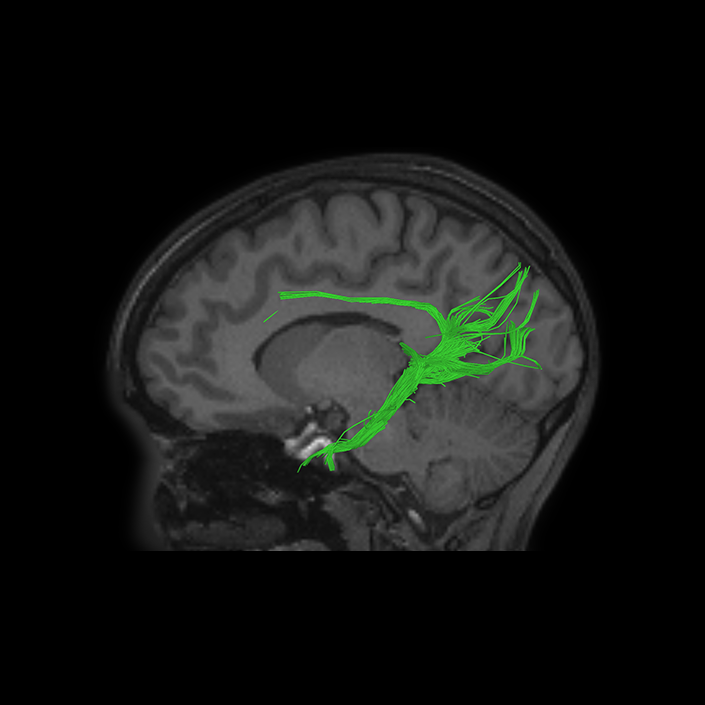
ᐅ SummaryArea 10r (10 rostral): part of medial superior frontal gyrus regions. Research suggests that it plays an important role in stimulus-oriented attention, indicating its importance in concentration and working memory. ᐅ Where is it?Area 10r (10 rostral) is located in the anterior inferior portion of the medial SFG ᐅ What are its borders?Area 10r borders area 10d and 10pp anteriorly and inferiorly, respectively. Its superior boundary is area p32 and its posterior boundary is area s32. ᐅ What are its functional connections?Area 10r demonstrates functional connectivity to areas 9p, 10d, and 8AD in the dorsolateral frontal lobe, areas 9m, a24, 10v, s32 and p32 in the medial frontal lobe, areas TGd, TE1a, STSva, PHA1, and the hippocampus in the temporal lobe, areas PGi and PGs in the lateral parietal lobe, and areas 7m, POS1, 31pv, 31pd, v23ab,and d23ab in the medial parietal lobe. ᐅ What are its white matter connections?Area 10r is connected to the contralateral hemisphere and cingulum. Connections to the contralateral hemisphere course through the genu of the corpus callosum to end at areas10r and 10v. Cingulum fibers project posteriorly from 10r to precuneus areas v23ab, POS1 and RSC. Local short association bundles connect with p32, 10d and 10v. White matter tracts in the right hemisphere have less consistent connections with the cingulum. ᐅ What is known about its function?The rostral portion of area 10 (or area 10r) is a newer parcellation of the pre-existing area 10. Research on this particular region suggests that area 10r plays an important role in stimulus-oriented attention, indicating its importance in concentration and working memory. |
|
A: lateral-medial
B: anterior-posterior
C: superior-inferior
DTI image |
Connectome Guide
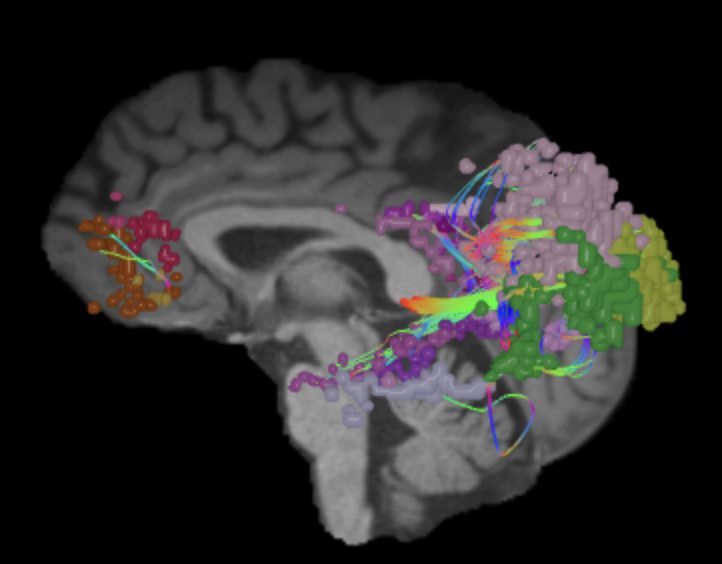
Default Mode Network (DMN)
Why we think this network is worth considering in decision making:
Hundreds of peer reviewed scientific papers about the default mode network have clearly defined this as one of the main cognitive and emotional regulation systems of the human cerebrum, and linked its dysfunction to impairment of brain function.
Evidence that this network is responsible for useful function in humans:
The default mode network is a distributed network of brain regions most active and connected during rest 1 however plays a vital role during information distribution while performing active tasks.2,3 DMN encompasses medial prefrontal cortex (mPFC), posterior cingulate cortex (PCC), precuneus, and lateral/medial temporal lobes.4 DTI studies identifies white matter tracts connecting the components of DMN.5 Multiple comparative anatomy studies with macaque monkeys provide reproducibility of the specific anatomy of the DMN. Tract tracing studies with non-human primates suggests that DMN connectivity is supported by anatomical connectivity.3,6 In addition, multiple human intracranial studies show task related suppression in neural activity in the distinct regions of the default mode network. Vanhaudenhuyse et al. (2010) has shown a exponential correlation between DMN connectivity integrity and the level of brain damaged patients, which suggested DMN as an indicator of patient’s consciousness.7 These result were repeated several times by multiple investigators.8,9,10
Consequences of damage to this network:
Several studies have shown DMN connectivity changes in several brain diseases including TBI,11 gliomas,12 schizophrenia,13 Alzheimer’s Disease,14 Parkinson’s Disease,15 epilepsy,16 and autism,17 indicating a link between disruption in this network and cognitive impairment.
Brain Tumors: In the case of glioma, it was reported that DMN connectivity alterations were also related with tumor grading such that in the low grade patients, DMN was lateralized to the hemisphere contralateral to tumor.18 Psychopathy is associated with changes in the default mode network.19 In a recent study, assessing the cognitive functions before and after surgical resection, Romero-Garcia et al. (2020) reported that memory recovery after brain tumor treatment is related to DMN impairment.20
Neurodegenerative diseases: Age related reductions is DMN activity and connectivity have been reported multiple times.21 Additionally, studies demonstrated a relationship between the cognitive decline and functional connectivity in the DMN22 Functional connectivity abnormalities in the DMN of patients with Alzheimer Disease has been consistently reported as well.23 Bucker et al., (2005), found a spatial overlap in the cortical regions that constitutes default mode network and amyloid deposition, this finding was repeated and extended to the connectivity patterns of the DMN.24,25,26 In addition, structural abnormalities, atrophy, were shown to be consistent with the spatial location of DMN regions.3,27 In Parkinson’s Disease, studies identified increased activation in the regions of DMN as well as abnormal connectivity pattern. In addition, visual hallucinations were also related to the abnormal activation in the DMN.28,29
Multiple studies reported a significant increase in DMN connectivity after treatment with medication in Alzheimer’s Disease30,31,32 and Parkinson’s Disease.33
Ischemic brain injury: Investigating focal lesions from stroke patients, Ferguson et al., (2019), identified a circuitry causing amnesia. Moreover, the topography of this circuitry spatially overlaps with the anatomical components of DMN, which are reported to be involved in AD.34
TBI: Bonnelle et al., (2012) showed DMN activation impairments correlating with behavioral measures after TBI, furthermore, another study showed DMN activity as the predictor of cognitive recovery in TBI.35,36 Several studies associated DMN changes with clinical outcomes. Caravasso et al., (2015) studied functional connectivity and neuropsychological changes in patients with severe acquired brain injury in a longitudinal experimental design to observe the effect of neurorehabilition in these measures. Their results suggested a close relationship between the strength of the connectivity changes in DMN and the clinical/cognitive recovery after the neurorehabilition. In other words, patients who showed strongest changes in DMN also showed more consistent cognitive recovery.37 A similar deficit and correlation was also shown in MS.38
Autism Spectrum disorders: Many studies demonstrated functional and structural connectivity impairments in the DMN in autism spectrum disorders.39 In line with these findings, volumetric gray matter increase in default mode network regions has been reported in male adolescents with autism.40
Schizophrenia: Several studies reported increased DMN activity/connectivity in the patients with schizophrenia , in individuals at ultra-risk for psychosis and unaffected siblings compared to healthy controls.41,42,43 Moreover, some studies demonstrated a relationship between DMN regulation and dopamine signaling with the evidence from pharmacological studies.44 In the case of schizophrenia, poorer clinical outcome was associated with reduced DMN connectivity.45
Radiation induced cognitive decline: DMN impairments were also identified with breast cancer patients treated with radiotherapy.46
ᐅ SummaryArea 31a (31 anterior): part of the subparietal gyrus. Considered a part of the dorsal posterior cingulate cortex, which is highly active during tasks that require an external focus, especially concerning visuospatial, and body orientation. Involved in working memory processing of place and body images; focusing on socially interacting objects vs randomly moving geometric shapes; and recognizing emotional faces over neutral objects. ᐅ Where is it?Area 31a (31 anterior) is found on the anterior half of the subparietal gyrus, directly posterior to the marginal sulcus. ᐅ What are its borders?Area 31a borders areas 31pd and 31pv and PCV posteriorly, and areas d23ab and 23d inferiorly. It has a long anterior and superior border with area 23c ᐅ What are its functional connections?Area 31a demonstrates functional connectivity to areas a9- 46v, p10p, 10d, 8AD, 8AV, 8C, s6-8 and i6-8 in the lateral frontal lobe, areas 8BM, p32, and d32 in the medial frontal lobe, areas PreS and TE1p in the temporal lobe, areas PGi, PGs, IP2, and IP1 in the lateral parietal lobe, and areas 23d, v23ab, d23ab, POS2, POS1, PCV, RSC, 7pm, 7m, 31pv, and 31pd in the medial parietal lobe. ᐅ What are its white matter connections?Area 31a is structurally connected to the cingulum and local parcellations of the precuneus. There are tracts that connect the contralateral hemisphere through the corpus callosum but this is inconsistent across individuals. The cingulum fibers project anteriorly from 31a with connections to the cingulate sulcus and superior frontal gyrus ending at areas p24, d32 and a24pr. Short association bundles project posterior to connect ᐅ What is known about its function?Area 31a is considered a part of the dorsal posterior cingulate cortex (dPCC) which is highly active during tasks that require an external focus, especially concerning visuospatial and body orientation. Task fMRIs indicate that this region is specifically involved in working memory processing of place and body images; focusing on socially interacting objects versus randomly moving geometric shapes; and recognizing emotional faces over neutral objects. |
|
A: lateral-medial
B: anterior-posterior
C: superior-inferior
DTI image |
ᐅ SummaryArea 31pd (31 posterior dorsal): part of the subparietal gyrus. Considered a part of the ventral posterior cingulate cortex, which is active during self-relevant tasks, including retrieval of semantic and episodic memories. Involved in working memory processing of body and face images; listening to stories vs answering arithmetic questions; and focusing on socially interacting objects vs randomly moving geometric shapes. ᐅ Where is it?Area 31pd (31 posterior dorsal) is found on the posterior superior portion of the subparietal gyrus. ᐅ What are its borders?Area 31pd borders area 31pv inferiorly, area 31a anteriorly, PCV superiorly, and area 7M posteriorly ᐅ What are its functional connections?Area 31pd demonstrates functional connectivity to areas a9-46v, 45, 47l, 47s, 10d, 8AD, 8AV, 8BL, 9a, and 9p in the lateral frontal lobe, areas SFL, 9m, 10r, 10v, and d32 in the medial frontal lobe, area TGd, STSva, STSvp, STSda, STSdp, TE1a, and the hippocampus in the temporal lobe, areas PGi, PGs, andIP2 in the lateral parietal lobe, and areas 23d, v23ab, d23ab, PCV, POS2, POS1, RSC, 7m, 31pv, and 31a in the medial parietal lobe. ᐅ What are its white matter connections?Area 31pd is structurally connected to the cingulum, contralateral hemisphere and local parcellations of the precuneus. The cingulum fibers project anteriorly from 31pd with variable connections along the cingulate sulcus and superior frontal gyrus. Connections project through the body of the corpus callosum to the contralateral precuneus to terminate at 31a, 7m and 31pd. Short association bundles are connected to 7m and PCV . ᐅ What is known about its function?Area 31pd is considered a part of the ventral posterior cingulate cortex (vPCC), which is active during self-relevant tasks, including retrieval of semantic and episodic memories. Task fMRI studies indicate that this region is specifically involved in working memory processing of body and face images; listening to stories versus answering arithmetic questions; and focusing on socially interacting objects versus randomly moving geometric shapes. |
|
A: lateral-medial
B: anterior-posterior
C: superior-inferior
DTI image |
ᐅ SummaryArea 31pv (31 posterior ventral): part of the subparietal gyrus. Considered a part of the ventral posterior cingulate cortex, which is active during self-relevant tasks, including retrieval of semantic and episodic memories. involved in working memory processing of body and face images; listening to stories over answering arithmetic questions; and recognizing emotional faces over neutral objects. ᐅ Where is it?Area 31pv (31 posterior ventral) is found on the posterior inferior subparietal gyrus where it spills across the cingulate sulcus onto the posterior cingulate gyrus. ᐅ What are its borders?Area 31pv borders area 31a anteriorly, and area 31pd superiorly. Its posterior border includes area 7m and area v23ab, and its inferior border is d23ab ᐅ What are its functional connections?Area 31pv demonstrates functional connectivity to areas 47l, 47s, p10p, 10d, 8AD, 8AV, 8BL, 8C, 9a, and 9p in the lateral frontal lobe, areas SFL, 9m, 10r, 10v, a24, p32, and d32 in the medial frontal lobe, area TGd, STSva, STSvp, TE1a, TE1m, PreS and the hippocampus in the temporal lobe, areas PGi, PGs, and PFm in the lateral parietal lobe, and areas 23d v23ab, d23ab, POS2, POS1, RSC, 7m, 31a, and 31pd in the medial parietal lobe. ᐅ What are its white matter connections?Area 31pv is structurally connected to the cingulum, contralateral hemisphere and local parcellations of the precuneus. The cingulum fibers project anteriorly from 31pv with variable connections along the cingulate sulcus and superior frontal gyrus. Connections project through the body of the corpus callosum to the contralateral precuneus to terminate at 31pv, 31a and 31pd. Short association bundles are connected to 23c, 23d, 31a, 31pd, 31pv and 7m. ᐅ What is known about its function?Area 31pv is considered a part of the ventral posterior cingulate cortex (vPCC), which is active during self-relevant tasks, including retrieval of semantic and episodic memories. Task fMRI studies indicate that this region is specifically involved in working memory processing of body and face images; listening to stories over answering arithmetic questions; and recognizing emotional faces over neutral objects. |
|
A: lateral-medial
B: anterior-posterior
C: superior-inferior
DTI image |
ᐅ SummaryArea a24 (anterior 24): part of anterior cingulate regions. Has been implicated as part of the affect division" of the anterior cingulate cortex ᐅ Where is it?Area a24 (anterior 24) is located in the anterior cingulate gyrus just anterior to the genu of the corpus callosum. ᐅ What are its borders?Area a24 borders area p24 superiorly and area 25 posteriorly. It abuts the genu of the corpus callosum. Its inferior border is s32 and its anterior border is made up of p32 and 9m. ᐅ What are its functional connections?Area a24 demonstrates functional connectivity to areas p24, d32, p32, s32, 10d, 10r, 9p, and 9m in the medial frontal lobe, area 8ad in the lateral frontal lobe, area TE1a in the temporal lobe, area PGs in the lateral parietal lobe, and areas RSC, 31pv, 31pd, 23d, d23ab, v23ab, 7m, and POS1 in the posterior cingulate areas. ᐅ What are its white matter connections?Area a24 is structurally connected to the cingulum. Cingulum fibers project anteriorly to end near the rostrum of the corpus callosum at 25. Posterior cingulum fibers end at the precuneus, near the splenium of the corpus callosum to area 31pv and 23d. Local short association bundles connect with p32 and 9m. ᐅ What is known about its function?Area a24 has been implicated as part of the "affect division" of the anterior cingulate cortex, and has been linked to analysis of internal and external states to play a role in emotional expression and motivation. |
|
A: lateral-medial
B: anterior-posterior
C: superior-inferior
DTI image |
ᐅ SummaryArea d23ab (dorsal 23 section a-b): part of the posterior cingulate cortex. Highly active during tasks that require an external focus, especially concerning visuospatial and body orientation. Involved in working memory processing of body images; listening to stories over answering arithmetic questions; and recognizing emotional faces over neutral objects. ᐅ Where is it?Area d23ab (dorsal 23 section a-b) is found on the posterior cingulate gyrus, just superior to the splenium of the corpus callosum. ᐅ What are its borders?Area d23ab borders area 23d anteriorly, and v23ab posteriorly. It has areas 31a, and 31pv as its superior border, and RSC as its inferior border. ᐅ What are its functional connections?Area d23ab demonstrates functional connectivity to areas a47r, p10p, i6-8, s6-8, 10d, 8AD, 8AV, 8BL, 8C, and 9p in the lateral frontal lobe, areas 8BM, 9m, 10r, a24, and d32 in the medial frontal lobe, area STSva, STSvp, TE1a, TE1m, TE1p, PreS and the hippocampus in the temporal lobe, areas IP1, PGi, PGs, and PFm in the lateral parietal lobe, and areas 23d, v23ab, POS2, POS1, RSC, 7m, 31a, 31pv, and 31pd in the medial parietal lobe. ᐅ What are its white matter connections?Area d23ab is structurally connected to the cingulum. The cingulum fibers project anteriorly from d23ab with connections to the anterior cingulate cortex and cingulate sulcus as it curves around the genu of the corpus callosum to terminate at a32pr and p24. Short association bundles project posterior to v23ab. ᐅ What is known about its function?Area d23ab is considered part of the dorsal posterior cingulate cortex (dPCC), which is highly active during tasks that require an external focus, especially concerning visuospatial and body orientation. Task fMRI studies indicate that this region is specifically involved in working memory processing of body images; listening to stories over answering arithmetic questions; and recognizing emotional faces over neutral objects. |
|
A: lateral-medial
B: anterior-posterior
C: superior-inferior
DTI image |
ᐅ SummaryArea IP1 (intraparietal 1): part of the lateral parietal lobe regions. Shows significant activation during mental arithmetic activities. Supports more complex parts of numeric and mathematical information processing. Shows greater activation in primary contrasts, when interpreting motor cues and when hearing a story compared to when hearing arithmetic problems. More active when individuals are processing faces than when processing shapes, and is less deactivated when hearing a story vs unrelated words. ᐅ Where is it?Area IP1 (Intraparietal 1) is found on the middle portion of the inferior bank of the intraparietal sulcus. ᐅ What are its borders?Area IP1 borders IP2 anteriorly and IP0 posteriorly. Its inferior border is formed by PFm and PGs, and its superior border is MIP and LIPv. ᐅ What are its functional connections?Area IP1 demonstrates functional connectivity to area 6a in the premotor regions, areas 33prime and 8BM in the middle cingulate regions, areas IFSa, IFSp, IFJp, a9-46v, p9-46v, 8AV, 8C, i6-8, a47r, and p47r in the lateral frontal lobe, areas TE1m, TE1p, PHT, and PreS in the temporal lobe, areas TE1p and PHT in the temporal lobe, areas PFm, PGs, IP0, IP2, AIP, MIP, and LIPd in the lateral parietal lobe, areas 7pm, 31a, d23ab, POS2, and RSC in the medial parietal lobe, and area V1 in the occipital lobe. ᐅ What are its white matter connections?Area IP1 is structurally connected with the superior longitudinal fasciculus (SLF). Connections with the SLF project anteriorly to the premotor cortex to end at 55b, FEF and PEF. Local short association fibers connect with PFm, LIPd, IP0, IPS1and PGs. White matter tracts in the right hemisphere have more inferior connections with the inferior frontal gyrus. ᐅ What is known about its function?Area IP1 shows significant activation during mental arithmetic activities, and, as part of the anterior IPS, supports more complex parts of numeric and mathematical information processing. Area IP1 shows greater activation in primary contrasts, when interpreting motor cues and when hearing a story compared to when hearing arithmetic problems. Relative to area IP2, area IP1 is more active when individuals are processing faces than when processing shapes, and is less deactivated when hearing a story versus unrelated words. |
|
A: lateral-medial
B: anterior-posterior
C: superior-inferior
DTI image |
ᐅ SummaryArea p32 (posterior 32): part of anterior cingulate regions. Has been shown to play a role in the emotional and cognitive integration of information during social interaction tasks, as well as play some role in error monitoring. ᐅ Where is it?Area p32 (posterior 32) is the central portion of the medial SFG. It borders the anterior bend of the callosal sulcus. ᐅ What are its borders?Area p32 borders area 9m superiorly, and 10d anteriorly. Its inferior borders include areas 10r and s32. Its posterior border includes a24 and a small portion of s32. ᐅ What are its functional connections?Area p32 demonstrates functional connectivity to areas d32, a24, p24, a32prime, and 10r in the medial frontal lobe, area 8AD in the lateral frontal lobe, and areas RSC, 31a, 31pv, POS2, and POS1 in the posterior cingulate areas. ᐅ What are its white matter connections?Area p32 is structurally connected to the contralateral hemisphere and cingulum. Connections to the contralateral hemisphere course through the genu of the corpus callosum to areas p32, 9m, 10d and 10v. Cingulum fibers project posteriorly to precuneus areas POS1, 31pv, RSC and v23ab. Local short association bundles connect with 9m, 10d, 10r and p24. ᐅ What is known about its function?Area p32 has been shown to play a role in the emotional and cognitive integration of information during social interaction tasks, as well as play some role in error monitoring. |
|
A: lateral-medial
B: anterior-posterior
C: superior-inferior
DTI image |
ᐅ SummaryArea POS1 (parieto-occipital sulcus 1): Activated during working memory processing of place images. This area also shows greater functional activity related to socially interacting objects vs randomly moving geometric shapes. It is suggested that this region is involved in scene-comprehension with the retrosplenial cortex. ᐅ Where is it?Area POS1 (Parieto-occipital sulcus 1) found on the anterior bank of the parieto- occipital sulcus, and makes up the inferior half of that bank. ᐅ What are its borders?Area POS1 borders area 7M, area d23ab, and RSC anteriorly, and DVT posteriorly. Its superior border is made of POS2, and its inferior border is made of ProS and RSC. ᐅ What are its functional connections?Area POS1 demonstrates functional connectivity to 8AD, i6- 8, 47m, 10d, 10v, and 10r in the lateral frontal lobe, areas 9m, a24, s32, p32, and d32 in the medial frontal lobe, areas STSva, TE1a, PHA1, PHA2, PHA3, PreS, and the hippocampus in the temporal lobe, areas PGi, PGp, and PGs in the lateral parietal lobe, and areas ProS, v23ab, d23ab, POS2, PCV, RSC, DVT, 7pm, 7m, 31a, 31pv, and 31pd in the medial parietal lobe. ᐅ What are its white matter connections?Area POS1 is structurally connected to the contralateral hemisphere and to anterior and parahippocampal cingulum projections. Anterior cingulum fibers have connections to the anterior cingulate cortex at a24, a24pr and p24. Posterior cingulum fibers curve around the splenium of the corpus callosum to end at the parahippocampal gyrus at area EC.Connections to contralateral V1 connect with FM. Short association bundles are connected to V1, V2, POS2 and V6. ᐅ What is known about its function?Task fMRI studies demonstrate that area POS1 is activated during working memory processing of place images. This area also shows greater functional activity related to socially interacting objects versus randomly moving geometric shapes. It is suggested that this region is involved in scene-comprehension with the RSC. |
|
A: lateral-medial
B: anterior-posterior
C: superior-inferior
DTI image |
ᐅ SummaryArea POS2 (parieto-occipital sulcus 2): Strong, coupled functional correlation with the retrosplenial cortex. Involved in working memory processing of place, body, tool, and face images; processing of visual cues instructing movement, and comparing featural dimensions of objects vs matching objects based on verbal classifications. ᐅ Where is it?Area POS2 (Parieto-occipital sulcus 2) is found on the anterior bank of the parieto- occipital sulcus, and makes up the superior half of that bank. ᐅ What are its borders?Area POS2 borders area 7M anteriorly and DVT posteriorly. Its superior border is made of areas 7pm and 7pl and its inferior border is made of POS1. ᐅ What are its functional connections?Area POS2 demonstrates functional connectivity to 46, 9- 46d, i6-8, 8C, p10p, and a10p in the lateral frontal lobe, areas 8BM, 9m, a24, p24, a32prime, p32, and d32 in the medial frontal lobe, areas IP1, IP2, PFm, PGp, and PGs in the lateral parietal lobe, and areas 23d, d23ab, POS1, PCV, RSC, DVT, 7am, 7pm, 7m, 31a, 31pv, and 31pd in the medial parietal lobe ᐅ What are its white matter connections?Area POS2 is structurally connected to local parcellations and the contralateral hemisphere. The white matter tracts from this parcellation are highly variable. Connections to the contralateral hemisphere travel with the forceps major fiber bundle to end at V1, though the termination of this tract is inconsistent. Short association bundles are connected to DVT, POS1, V2 and 7pm. ᐅ What is known about its function?Area POS2 has a strong, coupled functional correlation with the retrosplenial cortex (RSC). Task fMRI studies indicate that this region is specifically involved in working memory processing of place, body, tool, and face images; processing of visual cues instructing movement, and comparing featural dimensions of objects versus matching objects based on verbal classifications. |
|
A: lateral-medial
B: anterior-posterior
C: superior-inferior
DTI image |
ᐅ SummaryArea RSC (retrosplenial cortex): part of the posterior cingulate cortex. Primarily responsible for transitioning between allocentric (view-independent) spatial perspectives and egocentric (view-dependent) spatial perspectives. Implicated in spatial navigation, episodic memory, future planning, and imagination. Suspected of being involved in the retrieval of recent autobiographical information from memory. ᐅ Where is it?Area RSC (retrosplenial cortex) is a long thin area of posterior cingulate cortex which is immediately adjacent to the callosal sulcus wrapping around the splenium. Its length begins just superior to the midportion of the corpus callosum body, and it follows the cingulate sulcus until the bottom of the isthmus of the cingulum, where the parahippocampal gyrus begins. ᐅ What are its borders?Area RSC borders the corpus callosum along its entire inferior border. Its anterior border is with area 33prime, and its posteroinferior border is with the presubiculum. Its long superior border includes (from anterior to posterior): area 23d, area d23ab, area v23ab, POS1, and ProS. ᐅ What are its functional connections?Area RSC demonstrates functional connectivity to 9-46d, p10p, and 8AD in the lateral frontal lobe, areas 8BM, 9m, a24, p24, a32prime, p32, and d32 in the medial frontal lobe, areas IP1, IP2, PFm, and PGs in the lateral parietal lobe, and areas 23d, v23ab, d23ab, POS2, POS1, PCV, DVT, 7pm, 7m, 31a, 31pv, and 31pd in the medial parietal lobe. ᐅ What are its white matter connections?Area RSC is structurally connected to the cingulum. The cingulum fibers project both anteriorly and posteriorly from RSC, following the full length of the cingulate cortex. Posterior projections continue around the splenium of the corpus callosum ending at the parahippocampal gyrus at area EC. Anterior cingulum projections split near the genu of the corpus callosum to project superiorly to 10d and 9m, and inferiorly to 25. White matter tracts in the right hemisphere have more consistent connections with the precunues when compared to left. ᐅ What is known about its function?The retrosplenial cortex is primarily responsible for transitioning between allocentric (view-independent) spatial perspectives and egocentric (view-dependent) spatial perspectives. As such, the RSC is implicated in spatial navigation, episodic memory, future planning, and imagination. In addition, the RSC has been suspected of being involved in the retrieval of recent autobiographical information from memory. |
|
A: lateral-medial
B: anterior-posterior
C: superior-inferior
DTI image |
ᐅ SummaryArea PFm (parietal area F, part m): part of the lateral parietal lobe regions. Shows activation in nonspatial attention tasks, decision making when switching choices, rule change during visually guided attention, and reorientation. Also provide syntactical components to language processing, plays a role in attentional processing, and is activated in working memory, motor cue, and risk-related tasks. ᐅ Where is it?Area PFm (parietal area F, part m) is found on the anterior superior surface of the angular gyrus, and straddles the sulcus to lie on the posterior superior bank of the supramarginal gyrus. ᐅ What are its borders?Area PFm borders IP2 and IP1 superiorly, PF anteriorly, PSL and STV inferiorly, and PGI and PGS posteriorly. ᐅ What are its functional connections?Area PFm demonstrates functional connectivity to areas 8AV, 8AD, 8BL, 8C, s6-8, i6-8, a47r, p47r, a10p, p10p, 9a, a9-46v, p9-46v, and area 44 in the lateral frontal lobe, area d32 in the medial frontal lobe, area AVI in the insula, areas STSvp, TE1m, TE1p, and TE2a, in the temporal lobe, areas PGs, PGi, IP2, and IP1 in the lateral parietal lobe, and areas 7m, 7pm, POS2, 31a, 31pv, d23ab, 23d and RSC in the medial parietal lobe. ᐅ What are its white matter connections?Area PFm is structurally connected to the arcuate/SLF. Arcuate/SLF connections course anteriorly from PFm to 8C and 8BM, and inferiorly to middle temporal gyrus parcellations TE1a, TE1m, TE1p, STSva, STSvp and PHT. Local short association bundles connect with AIP, 7PC, IP1, IP2, LIPd, LIPv, PGi, PGs, 2 and 1 ᐅ What is known about its function?PFm shows activation in non-spatial attention tasks, decision making when switching choices, rule change during visually-guided attention, and reorientation. Intermediate regions of the inferior parietal lobule also provide syntactical components to language processing. This area also plays a role in attentional processing, and is activated in working memory, motor cue, and risk-related tasks. |
|
A: lateral-medial
B: anterior-posterior
C: superior-inferior
DTI image |
ᐅ SummaryArea PGi (parietal area G inferior): part of the lateral parietal lobe regions. Shows activity when individuals change their visuospatial attention from one area to another, and is a major node in the task-negative network, which mostly functions to redirect attention towards relevant stimuli. Relative to PGs, PGi is more active when processing faces compared to a body, is more active when listening to a story compared to unrelated words, and is more active when listening to a story vs answering arithmetic problems. ᐅ Where is it?Area PGi (parietal area G inferior) is found on the inferior surface of the angular gyrus. ᐅ What are its borders?Area PGi borders PGs superiorly, and PFm and STV anteriorly. Its inferior and its short posterior border is made up of TPOJ1, TPOJ2 and TPOJ3. ᐅ What are its functional connections?Area PGi demonstrates functional connectivity to areas 8AD, 8AV, 8BL, 8C, 47s, 47l, a47r, 44, 45, 10d, 9a, and 9p in the lateral frontal lobe, areas SFL, 9m, 10v, 10r, 8BM, and d32 in the medial frontal lobe, areas STSda, STSdp, STSva, STSvp, the hippocampus, TGd, TE1a, TE1m, TE1p, and TE2a, in the temporal lobe, areas PFm and PGs in the lateral parietal lobe, and areas 7m, POS1, 31a, 31pd, 31pv, d23ab, and v23ab in the medial parietal lobe. ᐅ What are its white matter connections?Area PGi is structurally connected to inferior parcellations of the occipitotemporal junction. Some individuals have connections to the arcuate/SLF with anterior projections to the premotor areas but this is inconsistent. Local short association bundles connect to the occipitotemporal junction at parcellations PHT, TE1p, STSvp, STSdp, TPOJ1 and TE1m. ᐅ What is known about its function?Area PGi is involved in many of the same functions as area PGs. This region shows activity when individuals change their visuospatial attention from one area to another, and is a major node in the task-negative network, which mostly functions to re-direct attention towards relevant stimuli. Relative to PGs, PGi is more active when processing faces compared to a body, is more active when listening to a story compared to un-related words, and is more active when listening to a story versus answering arithmetic problems. |
|
A: lateral-medial
B: anterior-posterior
C: superior-inferior
DTI image |
ᐅ SummaryArea PGs (parietal area G superior): part of the lateral parietal lobe regions. Shows activity when individuals change their visuospatial attention from one area to another. Specifically, it is involved in the response to biological motion. It is a major node in the task-negative network, which mostly functions to redirect attention towards relevant stimuli. Also relevant in number processing. ᐅ Where is it?Area PGs (parietal area G superior) is found on the superior surface of the angular gyrus. ᐅ What are its borders?Area PGs borders PFm anteriorly, PGi inferiorly, PGp posteriorly and IP1 and IP0 superiorly. ᐅ What are its functional connections?Area PGs demonstrates functional connectivity to areas 8AD, 8AV, 8BL, 8C, s6-8, i6-8, a47r, 47m, 10d, and 9p in the lateral frontal lobe, areas 9m, 10v, 10r, 8BM, a24, d32, and s32 in the medial frontal lobe, areas PHA1, PHA2, PreS, EC, the hippocampus, STSva, STSvp, TGd, TE1a, TE1m, TE1p, and TE2a, in the temporal lobe, areas PFm, PGi, and IP1 in the lateral parietal lobe, and areas 7m, 7pm, POS1, POS2, 31a, 31pd, 31pv, d23ab, v23ab, 23d and RSC in the medial parietal lobe. ᐅ What are its white matter connections?Area PGs is structurally connected to the arucate/SLF. Arcuate/SLF connections course anteriorly from PGs to inferior frontal sulcus parcellations IFJa, IFJp and IFSp, and inferiorly to occipitotemporal junction areas PHT, FST and TPOJ2. Local short association bundles connect with PFm, PGi, PGp, IP0, IP1, IP2 and MIP. ᐅ What is known about its function?Area PGs shows activity when individuals change their visuospatial attention from one area to another. Specifically, PGs is involved in the response to biological motion. It is a major node in the task-negative network, which mostly functions to re-direct attention toward relevant stimuli. This region is also relevant in number processing. Area PGs is superior to area PGi, but these regions are not differentiated well within the literature. |
|
A: lateral-medial
B: anterior-posterior
C: superior-inferior
DTI image |
ᐅ SummaryArea s32 (subcallosal 32): part of anterior cingulate regions. Heavily interconnected to other areas of the limbic system and thus plays a higher order role in emotional affect as well as reward expectation. ᐅ Where is it?Area s32 (subcallosal 32) lies in the subcallosal gyri. ᐅ What are its borders?Area s32 has a24, p32, and 10r as its anterior and superior borders. Area 10v and the OFC region are its inferior boundaries. Its posterior boundary is area 25. ᐅ What are its functional connections?Area s32 demonstrates functional connectivity to areas a24, 25, 10d, and 10r in the medial frontal lobe, area 8AD in the lateral frontal lobe, area PGs in the lateral parietal lobe, and areas v23ab and POS1 in the posterior cingulate areas. ᐅ What are its white matter connections?Area s32 is structurally connected to local parcellations. S32 has posterior connections to 25 and anterior connections to p32. ᐅ What is known about its function?Area s32 is heavily interconnected to other areas of the limbic system and thus plays a higher order role in emotional affect as well as reward expectation. |
|
A: lateral-medial
B: anterior-posterior
C: superior-inferior
DTI image |
ᐅ SummaryArea TPOJ3 (temporo-parieto-occipital junction 3): part of the lateral parietal lobe regions. Strongly activated when viewing faces compared to when viewing shapes. More active during working memory tasks, and less active during motor cue and shape recognition tasks. ᐅ Where is it?Area TPOJ3 (temporal-parietal-occipital junction 3) is found on the posterior inferior portion of the inferior parietal lobule. ᐅ What are its borders?Area TPOJ3 borders PGI superiorly, TPOJ2 anteriorly, MST, MT and LO3 inferiorly, and PGp posteriorly. ᐅ What are its functional connections?Area TPOJ3 demonstrates functional connectivity to area 2 in the sensory strip, areas 6a and FEF in the premotor areas, areas 5mv and 23c in the cingulate regions, areas OP4, PFcm, and STV in the insula opercular region, areas PHA2, PHA3, and TE2p in the temporal lobe, areas PGp, IP0, and 7PC in the lateral parietal lobe, areas 7AM, PCV, and DVT in the medial parietal lobe, area V2 in the medial occipital lobe, area V6 in the dorsal visual stream, and areas FST, PH, LO3, MST, TPOJ1 and TPOJ2 in the lateral occipital lobe. ᐅ What are its white matter connections?Area TPOJ3 is structurally connected with local parcellations and the inferior longitudinal fasciculus. Connections with the inferior longitudinal fasciculus project through the inferior temporal lobe to end at TF, TGv and PeEc areas. There are also projections to the premotor cortex but this is inconsistent across individuals. Local short association bundles connect with MT, MST, LO3, FST, TPOJ3, TE2p, PH and PGi. ᐅ What is known about its function?Area TPOJ3 is strongly activated when viewing faces compared to when viewing shapes. Relative to its anterior neighbor TPOJ2, area TPOJ3 is thinner, more active during working memory tasks, and less active during motor cue and shape recognition tasks. |
|
A: lateral-medial
B: anterior-posterior
C: superior-inferior
DTI image |
ᐅ SummaryArea v23ab (ventral 23 section a-b): part of the posterior cingulate cortex. Active during self-relevant tasks, including retrieval of semantic and episodic memories. Involved in working memory processing of body and face images; listening to stories over answering arithmetic questions; recognizing emotional faces over neutral objects; and focusing on socially interacting objects over randomly moving geometric shapes. ᐅ Where is it?Area v23ab (ventral 23 section a-b) is found on posterior most portion of the posterior cingulate area near the superior portion of the cingulate isthmus. ᐅ What are its borders?Area v23ab borders areas d23ab and 31pv anteriorly, and POS1 posteriorly. Area 7M is its superior neighbor, and RSC it its anterior-inferior border. ᐅ What are its functional connections?Area v23ab demonstrates functional connectivity to a47r, p10p, s6-8, 10d, 8AD, 8AV, 8BL, and 9p in the lateral frontal lobe, areas 8BM, 9m, 10r, 10v, a24, s32, and d32 in the medial frontal lobe, area STSva, TGd, TE1a, TE1m, PreS and the hippocampus in the temporal lobe, areas IP1, PGi, and PGs in the lateral parietal lobe, and areas d23ab, POS1, RSC, 7m, 31a, 31pv, and 31pd in the medial parietal lobe. ᐅ What are its white matter connections?Area v23ab is structurally connected to the cingulum. The cingulum fibers project anteriorly from v23ab with connections to the anterior cingulate cortex and cingulate sulcus as the fibers curve around the genu of the corpus callosum, these fibers end at a24, p24 and 32pr. The cingulum fibers continue wrapping around the genu with its connections splitting to project to the anterior pole of the frontal lobe at p32 and 10r, as well as following the rostrum of the corpus callosum to end at 25. Short association bundles are connected to POS1 and 7m. ᐅ What is known about its function?Area v23ab is considered a part of the ventral posterior cingulate cortex (vPCC), which is active during self-relevant tasks, including retrieval of semantic and episodic memories. Task fMRI studies indicate that this region is specifically involved in working memory processing of body and face images; listening to stories over answering arithmetic questions; recognizing emotional faces over neutral objects; and focusing on socially interacting objects over randomly moving geometric shapes. |
|
A: lateral-medial
B: anterior-posterior
C: superior-inferior
DTI image |
ᐅ SummaryArea PHA1 (parahippocampal area 1): part of the temporal lobe regons. Involved in visuospatial processing and episodic memory by processing contextual information. The anterior parahippocampal cortex is involved in encoding information without regard for stimulus category (scenes vs objects) or modality (word vs picture) and interfaces with the hippocampus, retrosplenial, and perirhinal memory systems, while the posterior parahippocampal cortex is involved with pictorial scene analysis, namely processing spatial features of visual scenes. ᐅ Where is it?Area PHA1 (parahippocampal area 1) is a long thin area on the medial portion of the parahippocampal gyrus. ᐅ What are its borders?Area PHA1 borders area PreS superiorly and PHA2 inferiorly. Its posterior border is VMV1 and its anterior border is EC. ᐅ What are its functional connections?Area PHA1 demonstrates functional connectivity to areas 10r and 8ad in the frontal lobe, areas PHA2, PHA3, VMV1 PreS, the hippocampus in the temporal lobe, and areas ProS, POS1, PGp, and PGs in the parietal lobe. ᐅ What are its white matter connections?Area PHA1 is connected to local parcellations. Local anterior fibers connect to PeEc. Local posterior fibers connect to PH. The exact terminations of the local connections are inconsistent across individuals. ᐅ What is known about its function?The parahippocampal cortex is involved in visuospatial processing and episodic memory by processing contextual information. The anterior parahippocampal cortex is involved in encoding information without regard for stimulus category (scenes vs objects) or modality (word vs picture) and interfaces with the hippocampus,retrosplenial, and perirhinal memory systems while the posterior parahippocampal cortex is involved with pictorial scene analysis, namely processing spatial features of visual scenes. Like both PHA2 and PHA3, PHA1 is activated in the PLACE-AVG contrast and deactivated in the FACE-AVG contrast, suggesting a role in place/scene recognition rather than face recognition. However, PHA1 is less deactivated in face recognition than PHA2 and PHA3 inferiorly. |
|
A: lateral-medial
B: anterior-posterior
C: superior-inferior
DTI image |
ᐅ SummaryArea PHA2 (parahippocampal area 2): part of the temporal lobe regions. Involved in visuospatial processing and episodic memory by processing contextual information. The anterior parahip- pocampal cortex is involved in encoding information without regard for stimulus category (scenes vs objects) or modality (word vs picture) and interfaces with the hippocampus, retrosplenial, and perirhinal memory systems, while the posterior parahippocampal cortex is involved with pictorial scene analysis, namely processing spatial features of visual scenes. ᐅ Where is it?Area PHA2 (parahippocampal area 2) is found on parahippocampal gyrus, primarily on its inferior surface, just adjacent to the collateral sulcus. ᐅ What are its borders?Area PHA2 is a long thin anterior posterior area between PHA1 superiorly and PHA3 inferiorly. It has a small porterior border with VMV2. ᐅ What are its functional connections?Area PHA2 demonstrates functional connectivity to areas 8AD, 47m, and i6-8 in the frontal lobe, areas PHA1, PHA3, PreS, and the hippocampus in the temporal lobe, areas ProS, 7pm, PCV, DVT, POS1, IP0, PGp, and PGs in the parietal lobe, and area TPOJ3 in the occipital lobe. ᐅ What are its white matter connections?Area PHA2 is structurally connected to the ILF. There are anterior projections from the ILF that terminate at PeEc. The posterior terminations of the ILF terminate at V1, V2 and V3. The exact terminations of the local connections are inconsistent across individuals. ᐅ What is known about its function?The parahippocampal cortex is involved in visuospatial processing and episodic memory by processing contextual information. The anterior parahippocampal cortex is involved in encoding information without regard for stimulus category (scenes vs objects) or modality (word vs picture) and interfaces with the hippocampus, retrosplenial, and perirhinal memory systems while the posterior parahippocampal cortex is involved with pictorial scene analysis, namely processing spatial features of visual scenes. Like both PHA1 and PHA3, PHA2 is activated in the PLACE-AVG contrast and deactivated in the FACE-AVG contrast, suggesting a role in place/scene recognition rather than face recognition. Area PHA2 is more deactivated than PHA1 in facial recognition tasks. |
|
A: lateral-medial
B: anterior-posterior
C: superior-inferior
DTI image |
ᐅ SummaryArea PHA3 (parahippocampal area 3): part of the temporal lobe regions. Involved in visuospatial processing and episodic memory by processing contextual information. The anterior parahippocampal cortex is involved in encoding information without regard for stimulus category (scenes vs objects) or modality (word vs picture) and interfaces with the hippocampus, retrosplenial, and perirhinal memory systems, while the posterior parahippocampal cortex is involved with pictorial scene analysis, namely processing spatial features of visual scenes. ᐅ Where is it?Area PHA3 (parahippocampal area 3) is located in the parahippocampal gyrus, primarily inside the collateral sulcus ᐅ What are its borders?Area PHA3 borders PHA2 superiorly, and VVC (ventral visual complex) and TF inferiorly. Its posterior border is with VMV2 and VMV3, and its anterior border is PeEC. ᐅ What are its functional connections?Area PHA3 demonstrates functional connectivity to areas 6a and IFSa in the frontal lobe, areas PHT, PHA1, PHA2, VMV2 and PeEc in the temporal lobe, areas 23c, 7pl, 7am, 7pm, PCV, DVT POS1, MIP, LIPd, AIP, PGp, and PFt in the parietal lobe, and areas TPOJ3 and VVC in the occipital lobe. ᐅ What are its white matter connections?Area PHA3 is structurally connected to the ILF. There are anterior projections from the ILF that terminate at TGd. The posterior terminations of the ILF terminate at VVC and FFC. There are fibers that run parallel to the ILF and terminate at the occipital lobe at V1, V2 and V6. The exact terminations of the local connections are inconsistent across individuals. ᐅ What is known about its function?The parahippocampal cortex is involved in visuospatial processing and episodic memory by processing contextual information. The anterior parahippocampal cortex is involved in encoding information without regard for stimulus category (scenes vs objects) or modality (word vs picture) and interfaces with the hippocampus, retrosplenial, and perirhinal memory systems while the posterior parahippocampal cortex is involved with pictorial scene analysis, namely processing spatial features of visual scenes. Relative to PHA2, area PHA3 demonstrates greater activity in tool-related recognition tasks. |
|
A: lateral-medial
B: anterior-posterior
C: superior-inferior
DTI image |
ᐅ SummaryArea PreS: (presubiculum) part of the medial temporal areas. Region of the hippocampus that primate studies suggest is involved in the processing of spatial information. Demonstrates less activity during tasks related to working memory, language processing, and theory of mind. Relative to PHA1, PreS shows greater activity during motor tasks. ᐅ Where is it?Area PreS is found on the posterior superior surface of the parahippocampal gyrus. ᐅ What are its borders?Area PreS borders the hippocampus medially, and the entrorhinal cortex anteriorly. Its posterior border is made up of RSC (retrosplenial cortex) and the ProS (prostriate region) (which are discussed in other sections. PHA1 is its inferior border ᐅ What are its functional connections?Area PreS demonstrates functional connectivity areas 8AD and i6-8 in the frontal lobe, areas PHA1, PHA2, and the hippocampus in the temporal lobe, areas RSC, Pros, d23ab, v23ab, 31a, 31pv, 7m, 7pm, POS1, POS2, IP1, and PGs in the parietal lobe, and area V1 in the occipital lobe ᐅ What are its white matter connections?Area PreS is structurally connected to the cingulum, precuneus and occipital lobe. Cingulum projections run superior to the corpus callosum to end at anterior cingulate cortex and frontal lobe parcellations a24, 9m, 10d and p32. There arePreS fibers that project posteriorly to end at occipital and precuneus areas V1, V2, V6, POS1, POS2 and 7m. Local short association fibers are connected to EC and PeEc. ᐅ What is known about its function?The presubiculum lies medial to the subiculum - a region of the hippocampus which primate studies suggest is involved in the processing of spatial information. Area PreS contains more myelin than the hippocampal cortex, and relative to PHA1 inferiorly, contains more myelin, is thinner, and demonstrates less activity during tasks related to working memory, language processing, and theory of mind. Relative to PHA1, PreS shows greater activity during motor tasks. |
|
A: lateral-medial
B: anterior-posterior
C: superior-inferior
DTI image |
Reference list
- Raichle E. The Brain’s Default Mode Network. Annual Review of Neuroscience, 2015; 38(1), 433–447. https://doi.org/10.1146/annurev-neuro-071013-014030
- Anticevic A, Cole W, Murray D, Corlett R, Wang J, Krystal H. The role of default network deactivation in cognition and disease. In Trends in Cognitive Sciences, 2012;16(12), 584-594. NIH Public Access. https://doi.org/10.1016/j.tics.2012.10.008
- Buckner L, Andrews-Hanna R, Schacter L. The brain’s default network: Anatomy, function, and relevance to disease. Annals of the New York Academy of Sciences, 2008;1124(April), 1–38. https://doi.org/10.1196/annals.1440.011
- Raichle E, Snyder Z. A default mode of brain function: A brief history of an evolving idea. NeuroImage 2007; 37(4), 1083-1090. Academic Press Inc. https://doi.org/10.1016/j.neuroimage.2007.02.041
- van den Heuvel P, Mandl W, Kahn S, Hulshoff E. Functionally linked resting-state networks reflect the underlying structural connectivity architecture of the human brain. Human Brain Mapping, 2009;30(10), 3127–3141. https://doi.org/10.1002/hbm.20737
- Buckner L, DiNicola M. (2019). The brain’s default network: updated anatomy, physiology and evolving insights. Nature Reviews Neuroscience, 2019;20(10), 593-608. Nature Research. https://doi.org/10.1038/s41583-019-0212-7
- Vanhaudenhuyse A, Noirhomme Q, Tshibanda F, Bruno A, Boveroux P, Schnakers C, Soddu A, Perlbarg V, Ledoux D, Brichant F, Moonen G, Maquet P, Greicius D, Laureys S, Boly M. Default network connectivity reflects the level of consciousness in non-communicative brain-damaged patients. Brain, 2010;133(1), 161–171. https://doi.org/10.1093/brain/awp313
- Boly M, Tshibanda L, Vanhaudenhuyse A, Noirhomme Q, Schnakers C, Ledoux D, Boveroux P, Garweg C, Lambermont B, Phillips C, Luxen A, Moonen G, Bassetti C, Maquet P, Laureys S. Functional connectivity in the default network during resting state is preserved in a vegetative but not in a brain dead patient. Human Brain Mapping, 2009;30(8), 2393–2400. https://doi.org/10.1002/hbm.20672
- Norton L, Hutchison M, Young B, Lee H, Sharpe D, Mirsattari M. Disruptions of functional connectivity in the default mode network of comatose patients. Neurology, 2012;78(3), 175–181. https://doi.org/10.1212/WNL.0b013e31823fcd61
- Soddu A, Vanhaudenhuyse A, Bahri A, Bruno A, Boly M, Demertzi A, Tshibanda F, Phillips C, Stanziano M, Ovadia-Caro S, Nir Y, Maquet P, Papa M, Malach R, Laureys S, Noirhomme Q. Identifying the default-mode component in spatial IC analyses of patients with disorders of consciousness. Human Brain Mapping, 2012;33(4), 778–796. https://doi.org/10.1002/hbm.21249
- Sharp J, Beckmann F, Greenwood R, Kinnunen M, Bonnelle V, De Boissezon X, Powell H, Counsell J, Patel C, Leech R. Default mode network functional and structural connectivity after traumatic brain injury. Brain, 2011; 134(8), 2233–2247. https://doi.org/10.1093/brain/awr175
- Ghumman S, Fortin D, Noel-Lamy M, Cunnane C, Whittingstall K. Exploratory study of the effect of brain tumors on the default mode network. Journal of Neuro-Oncology, 2016; 128(3), 437–444. https://doi.org/10.1007/s11060-016-2129-6
- Whitfield-Gabrieli S, Thermenos W, Milanovic S, Tsuang T, Faraone V, McCarley W, Shenton E, Green I, Nieto-Castanon A, LaViolette P, Wojcik J, Gabrieli E, Seidman J. Hyperactivity and hyperconnectivity of the default network in schizophrenia and in first-degree relatives of persons with schizophrenia. Proceedings of the National Academy of Sciences of the United States of America, 2009;106(4), 1279–1284. https://doi.org/10.1073/pnas.0809141106
- Supekar, Kaustubh, Menon V, Rubin D, Musen M, Greicius D. Network analysis of intrinsic functional brain connectivity in Alzheimer’s disease. PLoS Computational Biology, 2008;4(6). https://doi.org/10.1371/journal.pcbi.1000100
- Putcha D, Ross S, Cronin-Golomb A, Janes C, Stern E. (2015). Altered intrinsic functional coupling between core neurocognitive networks in Parkinson’s disease. NeuroImage: Clinical, 2015;7, 449–455. https://doi.org/10.1016/j.nicl.2015.01.012
- Hsiao J, Yu Y, Chen T, Kwan Y, Chen C, Yen J, Yiu H, Shih H, Lin Y. Increased intrinsic connectivity of the default mode network in temporal lobe epilepsy: Evidence from resting-state MEG recordings. PLoS ONE, 2015;10(6). https://doi.org/10.1371/journal.pone.0128787
- Castelli F, Frith C, Happé F, Frith U. Autism, asperger syndrome and brain mechanisms for the attribution of mental states to animated shapes. Brain, 2002;125(8), 1839–1849. https://doi.org/10.1093/brain/awf189
- Esposito R, Mattei A, Briganti C, Romani L, Tartaro A, Caulo M. Modifications of default-mode network connectivity in patients with cerebral glioma. PLoS ONE, 2012;7(7). https://doi.org/10.1371/journal.pone.0040231
- Sethi A, Gregory S, Dell’Acqua F, Periche Thomas E, Simmons A, Murphy M, Hodgins S, Blackwood J, Craig C. Emotional detachment in psychopathy: Involvement of dorsal default-mode connections. Cortex, 2015;62, 11–19. https://doi.org/10.1016/j.cortex.2014.07.018
- Romero-Garcia R, Suckling J, Owen M, Assem M, Sinha R, Coelho P, Woodberry E, Price J, Burke A, Santarius T, Erez Y, Hart G. Memory recovery is related to default mode network impairment and neurite density during brain tumours treatment. medRxiv, 2020;19008581. medRxiv. https://doi.org/10.1101/19008581
- Desgranges B, Mevel K, Chételat G, Eustache F. The default mode network in healthy aging and Alzheimer’s disease. In International Journal of Alzheimer’s Disease, 2011. https://doi.org/10.4061/2011/535816
- Grieder M, Wang J, Dierks T, Wahlund O, Jann K.Default Mode Network Complexity and Cognitive Decline in Mild Alzheimer’s Disease. Frontiers in Neuroscience, 2018;12. https://doi.org/10.3389/fnins.2018.00770
- Gili T, Cercignani M, Serra L, Perri R, Giove F, Maraviglia B, Caltagirone C, Bozzali M. Regional brain atrophy and functional disconnection across Alzheimer’s disease evolution. Journal of Neurology, Neurosurgery and Psychiatry, 2011; 82(1), 58–66. https://doi.org/10.1136/jnnp.2009.199935
- Buckner L, Snyder Z, Shannon J, LaRossa G, Sachs R, Fotenos F, Sheline I, Klunk E, Mathis A, Morris C, Mintun A. Molecular, structural, and functional characterization of Alzheimer’s disease: Evidence for a relationship between default activity, amyloid, and memory. Journal of Neuroscience, 2015;25(34), 7709–7717. https://doi.org/10.1523/JNEUROSCI.2177-05.2005
- Hedden T, Van Dijk A, Becker A, Mehta A, Sperling A, Johnson A, Buckner L. (2009). Disruption of functional connectivity in clinically normal older adults harboring amyloid burden. Journal of Neuroscience, 2009;29(40), 12686–12694. https://doi.org/10.1523/JNEUROSCI.3189-09.2009
- Mormino C, Smiljic A, Hayenga O, Onami S, Greicius D, Rabinovici D, Janabi M, Baker L, Yen I, Madison M, Miller L, Jagust J. Relationships between beta-amyloid and functional connectivity in different components of the default mode network in aging. Cerebral Cortex, 2011; 21(10), 2399–2407. https://doi.org/10.1093/cercor/bhr025
- Scahill I, Schott M, Stevens M, Rossor N, Fox C. Mapping the evolution of regional atrophy in Alzheimer’s disease: Unbiased analysis of fluid-registered serial MRI. Proceedings of the National Academy of Sciences of the United States of America, 2002; 99(7), 4703–4707. https://doi.org/10.1073/pnas.052587399
- Franciotii R, Delli Pizzi S, Perfetti B, Tartaro A, Bonanni L, Thomas A, Weis L, Biundo R, Antonini A, Onofrj M. Default mode network links to visual hallucinations: A comparison between Parkinson’s disease and multiple system atrophy. Movement Disorders, 2015; 30(9), 1237–1247. https://doi.org/10.1002/mds.26285
- Putcha D., Ross, R. S., Cronin-Golomb, A., Janes, A. C., & Stern, C. E. (2015). Altered intrinsic functional coupling between core neurocognitive networks in Parkinson’s disease. NeuroImage: Clinical, 7, 449–455. https://doi.org/10.1016/j.nicl.2015.01.012
- Goveas S, Xie C, Ward D, Wu Z, Li W, Franczak M, Jones L, Antuono G, Li J. Recovery of hippocampal network connectivity correlates with cognitive improvement in mild Alzheimer’s disease patients treated with donepezil assessed by resting-state fMRI. Journal of Magnetic Resonance Imaging, 2011;34(4), 764–773. https://doi.org/10.1002/jmri.22662
- Li W, Antuono G, Xie C, Chen G, Jones L, Ward D, Franczak B, Goveas S, Li J. Changes in regional cerebral blood flow and functional connectivity in the cholinergic pathway associated with cognitive performance in subjects with mild Alzheimer’s disease after 12-week donepezil treatment. NeuroImage, 2012;60(2), 1083–1091. https://doi.org/10.1016/j.neuroimage.2011.12.077
- Lorenzi M, Beltramello A, Mercuri B, Canu E, Zoccatelli G, Pizzini B, Alessandrini F, Cotelli M, Rosini S, Costardi D, Caltagirone C, Frisoni B. Effect of memantine on resting state default mode network activity in Alzheimer's disease. Drugs and Aging, 2011;28(3), 205-217. https://doi.org/10.2165/11586440-000000000-00000
- Delaveau P, Salgado-Pineda P, Fossati P, Witjas T, Azulay P, Blin O. Dopaminergic modulation of the default mode network in Parkinson’s disease. European Neuropsychopharmacology, 2010;20(11), 784–792. https://doi.org/10.1016/j.euroneuro.2010.07.001
- Ferguson A, Lim C, Cooke D, Darby R, Wu O, Rost S, Corbetta M, Grafman J, Fox D. A human memory circuit derived from brain lesions causing amnesia. Nature Communications, 2019;10(1). https://doi.org/10.1038/s41467-019-11353-z
- Bonnelle V, Ham E, Leech R, Kinnunen M, Mehta A, Greenwood J, Sharp J. Salience network integrity predicts default mode network function after traumatic brain injury. Proceedings of the National Academy of Sciences, 2012;109(12), 4690–4695. https://doi.org/10.1073/pnas.1113455109
- Zhou, Yongxia, Milham P, Lui W, Miles L, Reaume J, Sodickson K, Grossman I, Ge Y. Default-mode network disruption in mild traumatic brain injury. Radiology, 2012;265(3), 882–892. https://doi.org/10.1148/radiol.12120748
- Falletta Caravasso C, De Pasquale F, Ciurli P, Catani S, Formisano R, Sabatini U. The Default Mode Network Connectivity Predicts Cognitive Recovery in Severe Acquired Brain Injured Patients: A Longitudinal Study. Journal of Neurotrauma, 2016;33(13), 1247–1262. https://doi.org/10.1089/neu.2015.4003
- Rocca A, Valsasina P, Absinta M, Riccitelli G, Rodegher E, Misci P, Rossi P, Falini A, Comi G, Filippi M. Default-mode network dysfunction and cognitive impairment in progressive MS. Neurology, 2010;74(16), 1252–1259. https://doi.org/10.1212/WNL.0b013e3181d9ed91
- Padmanabhan A, Lynch J, Schaer M, Menon V, Quarry R. The Default Mode Network in Autism HHS Public Access. Biol Psychiatry Cogn Neurosci Neuroimaging, 2017;2(6), 476–486. https://doi.org/10.1016/j.bpsc
- Waiter D, Williams G, Murray D, Gilchrist A, Perrett I, Whiten A. A voxel-based investigation of brain structure in male adolescents with autistic spectrum disorder. NeuroImage, 2004;22(2), 619–625. https://doi.org/10.1016/j.neuroimage.2004.02.029
- Liu H, Kaneko Y, Ouyang X, Li L, Hao Y, Chen H, Jiang T, Zhou Y, Liu Z. Schizophrenic patients and their unaffected siblings share increased resting-state connectivity in the task-negative network but not its anticorrelated task-positive network. Schizophrenia Bulletin, 2012;38(2), 285–294. https://doi.org/10.1093/schbul/sbq074
- Shim G, Oh S, Jung H, Jang H, Choi H, Kim E, Park Y, Choi S, Jung H, Kwon S. Altered resting-state connectivity in subjects at ultra-high risk for psychosis: An fMRI study. Behavioral and Brain Functions, 2010;6. https://doi.org/10.1186/1744-9081-6-58
- Zhou, Yuan, Liang M, Tian L, Wang K, Hao Y, Liu H, Liu Z, Jiang T. Functional disintegration in paranoid schizophrenia using resting-state fMRI. Schizophrenia Research, 2007;97(1–3), 194–205. https://doi.org/10.1016/j.schres.2007.05.029
- Nagano-Saito A, Liu J, Doyon J, Dagher A. Dopamine modulates default mode network deactivation in elderly individuals during the Tower of London task. Neuroscience Letters, 2009;458(1), 1–5. https://doi.org/10.1016/j.neulet.2009.04.025
- Lee H, Lee K, Park K, Kim E, Ryu S. Default mode network connectivity is associated with long-term clinical outcome in patients with schizophrenia. NeuroImage: Clinical, 2019;22(December 2018), 101805. https://doi.org/10.1016/j.nicl.2019.101805
- Miao H, Chen X, Yan Y, He X, Hu S, Kong J, Wu M, Wei Y, Zhou Y, Wang L, Wang K, Qiu B. Functional connectivity change of brain default mode network in breast cancer patients after chemotherapy. Neuroradiology, 2016;58(9), 921–928. https://doi.org/10.1007/s00234-016-1708-8